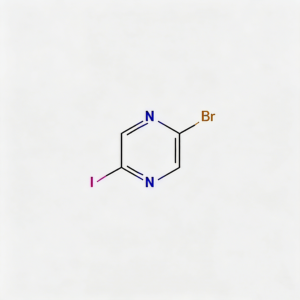
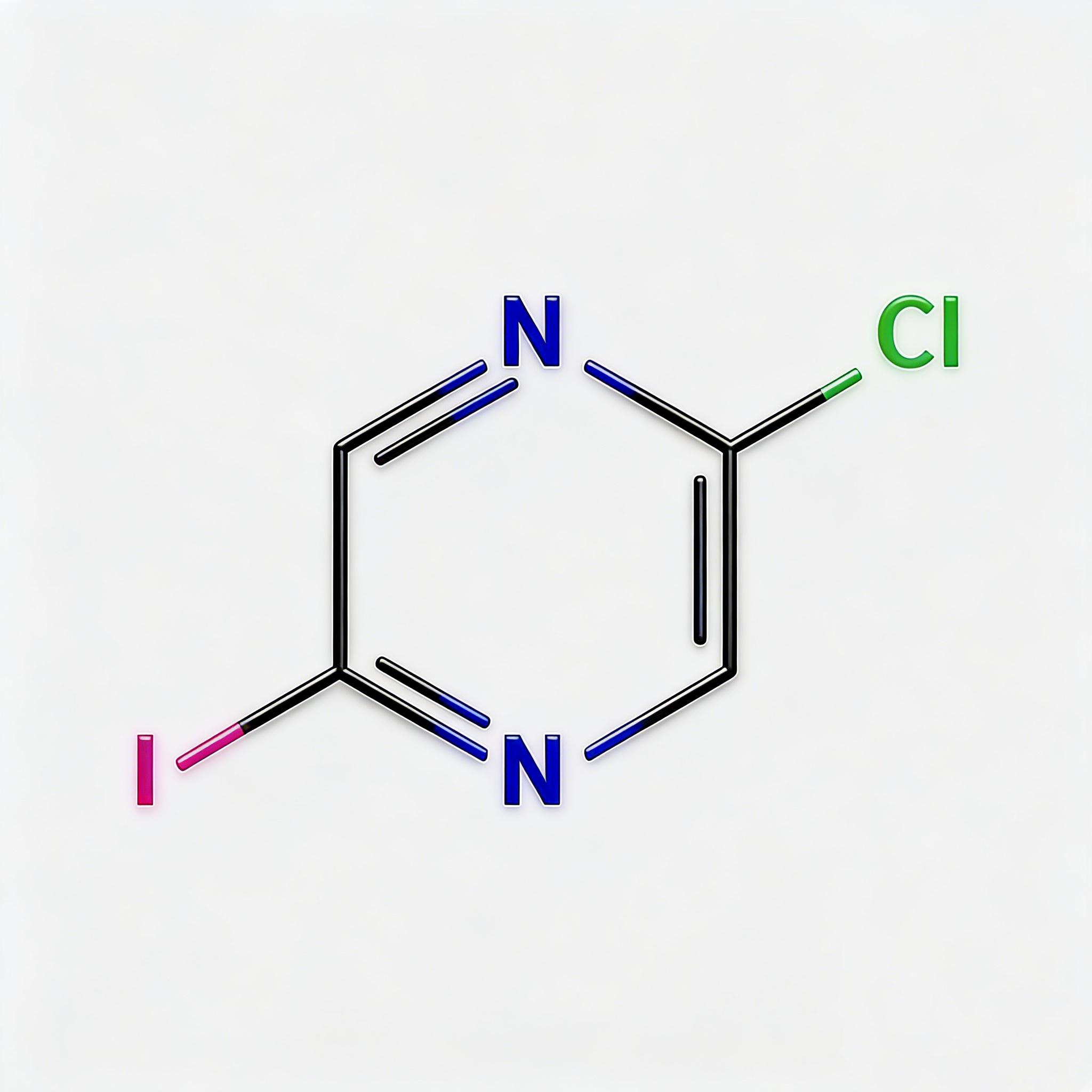
2-Bromo-5-iodopyrazine
2-Bromo-5-iodopyrazine (CAS 622392-04-5) is a strategically bifunctional halogenated pyrazine scaffold engineered for sequential cross-coupling reactions, pharmaceutical intermediate synthesis, and materials science development. This molecule integrates an electron-deficient pyrazine core—a six-membered aromatic diazine ring with nitrogen atoms at para positions (1,4)—with a bromine substituent at the C2 position and an iodine atom at the C5 position. The dual halogen architecture with differentiated reactivity (I > Br) enables chemoselective functionalization, allowing researchers to introduce distinct substituents at each position through controlled Pd-catalyzed cross-coupling (Suzuki, Stille, Negishi, Sonogashira). The 2,5-substitution pattern creates a symmetric geometry across the ring center, while the iodine handle offers faster oxidative addition kinetics compared to the bromine site. This intermediate is particularly valuable for constructing asymmetric ligands, fused heterocycles (pyrazino-fused systems), kinase inhibitor cores, and conjugated materials where stepwise derivatization is critical. Manufactured with purity ≥98% under controlled conditions, it supports drug discovery programs requiring modular access to differentially halogenated electron-deficient heterocycles.
Property
|
Product Name
|
2-Bromo-5-iodopyrazine
|
|
CAS Number
|
622392-04-5
|
|
Chinese Name
|
2-溴 -5-碘吡嗪
|
|
Synonyms
|
5-Iodo-2-bromopyrazine; Pyrazine, 2-bromo-5-iodo-; C₄H₂BrIN₂ heterocycle; Br-I-Pyrazine
|
|
Chinese Synonyms
|
5-碘 -2-溴吡嗪; 2-溴 -5-碘代吡嗪; 溴碘吡嗪; 2,5-位卤代吡嗪
|
|
Molecular Formula
|
C₄H₂BrIN₂
|
|
Molecular Weight
|
284.88 g/mol
|
|
Purity
|
≥ 98% (HPLC/GC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Pyrazine Derivatives / Mixed Dihalo heterocycles
|
|
Appearance
|
White to Off-white to Light Yellow Crystalline Powder
|
|
Melting Point
|
~86-90°C (estimated, literature reference recommended)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed in amber glass under inert atmosphere (nitrogen). Protect from light to avoid iodine degradation and from strong bases to avoid premature halide displacement.
|
|
Shelf Life
|
18-24 months under recommended storage conditions (monitor for color darkening due to iodine)
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements (amber glass recommended)
|
|
Applications
|
Kinase inhibitor synthesis, sequential cross-coupling reactions (chemoselective functionalization), asymmetric ligand construction, heterocyclic library construction (pyrazino-fused systems), conjugated material precursor, agrochemical intermediate
|
|
Reactivity
|
Suitable for selective Pd-catalyzed cross-coupling (I reacts faster than Br), nucleophilic aromatic substitution, lithiation, electrophilic aromatic substitution at C3/C6, and stepwise derivatization
|
|
Special Note
|
✅ Chemoselective Dihalo Scaffold—Iodine (C5) reacts faster than Bromine (C2), enabling sequential functionalization. Light-Sensitive—iodine substituent requires amber storage. Distinct from symmetric dihalides (e.g., 2,5-dichloro). Part of our halogenated pyrazine series.
|