You are here:
- Home
- Aromatic hydrocarbons
- Benzyl carbamate
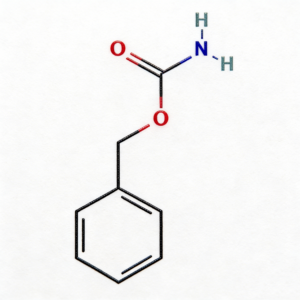
Benzyl carbamate
Benzyl carbamate (CAS 621-84-1) is a high-purity carbamate ester intermediate widely used in pharmaceutical synthesis and organic chemistry research. This compound serves as a versatile protecting group reagent and building block for the development of complex organic molecules and active pharmaceutical ingredients (APIs). Featuring a benzyloxycarbonyl (Cbz/Z) functional group, it exhibits excellent stability and reactivity in peptide synthesis, amine protection/deprotection chemistry, and various nucleophilic substitution reactions. Our product is manufactured under strict quality control standards, ensuring a purity of ≥98%, making it ideal for pharmaceutical manufacturing, medicinal chemistry research, and scale-up production. This compound is particularly valuable in the synthesis of peptide-based pharmaceuticals, amino acid derivatives, and fine chemicals requiring carbamate functionality.
Property
|
Product Name
|
Benzyl carbamate
|
|
CAS Number
|
621-84-1
|
|
Chinese Name
|
氨基甲酸苄酯
|
|
Synonyms
|
Cbz-NH₂; Z-NH₂; Carbamic acid, phenylmethyl ester; Phenylmethyl carbamate; Benzyl urethane; N-Cbz-ammonia; Benzyloxycarbonyl amine; Cbz-amine; Z-amine
|
|
Chinese Synonyms
|
苄氧羰基胺; Cbz-胺; Z-胺; 氨基甲酸苯甲酯; 苄基氨基甲酸酯
|
|
Molecular Formula
|
C₈H₉NO₂
|
|
Molecular Weight
|
151.16 g/mol
|
|
Purity
|
≥ 98%
|
|
Product Category
|
Pharmaceutical Intermediates / Carbamates / Protecting Group Reagents
|
|
Appearance
|
White to Off-white Crystalline Powder
|
|
Melting Point
|
88-90°C
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
1kg/foil bag, 5kg/drum, 25kg/drum, or customized according to customer requirements
|
|
Applications
|
Pharmaceutical intermediates, peptide synthesis, amine protection chemistry, organic synthesis, API manufacturing, fine chemical production
|
|
Reactivity
|
Suitable for Cbz protection of amines, peptide coupling reactions, nucleophilic substitution, and deprotection under hydrogenolysis conditions
|
|
Special Note
|
Widely used protecting group reagent in peptide and protein chemistry
|