You are here:
- Home
- Aromatic hydrocarbons
- 2,6-Dibromopyridine
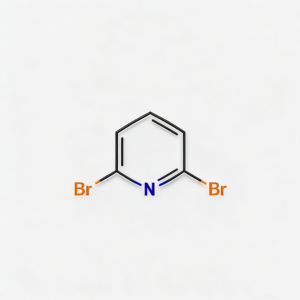
2,6-Dibromopyridine
2,6-Dibromopyridine (CAS 626-05-1) is a strategically symmetric halogenated heterocyclic scaffold engineered for cross-coupling reactions, ligand synthesis, and pharmaceutical intermediate development. This molecule integrates a pyridine core—a nitrogen-containing six-membered aromatic ring—with two bromine substituents at the C2 and C6 positions, creating a symmetric C₂ᵥ architecture. The dual bromine handles are activated by the adjacent ring nitrogen, enabling efficient sequential or simultaneous Pd-catalyzed cross-coupling (Suzuki, Stille, Negishi, Sonogashira) to introduce diverse aryl, alkyl, or heteroaryl groups. The symmetric substitution pattern makes it a privileged precursor for constructing terpyridine ligands, conjugated polymers, and bis-heterocyclic kinase inhibitors where precise spatial arrangement of binding motifs is critical. Compared to mono-brominated or asymmetrically substituted pyridines, 2,6-dibromopyridine offers streamlined synthetic routes to C2-symmetric structures and enhanced binding affinity through chelation effects. Manufactured with purity ≥98% under controlled conditions, it supports drug discovery, materials science, and coordination chemistry programs requiring modular access to symmetrically functionalized pyridine architectures.
Property
|
Product Name
|
2,6-Dibromopyridine
|
|
CAS Number
|
626-05-1
|
|
Chinese Name
|
2,6-二溴吡啶
|
|
Synonyms
|
Pyridine, 2,6-dibromo-; 2,6-Dibromopyridine; C₅H₃Br₂N heterocycle; DBP
|
|
Chinese Synonyms
|
2,6-二溴吡啶; 二溴吡啶; 2,6-二溴氮苯; 对称二溴吡啶
|
|
Molecular Formula
|
C₅H₃Br₂N
|
|
Molecular Weight
|
236.89 g/mol
|
|
Purity
|
≥ 98% (GC/HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Pyridine Derivatives / Dihalo heterocycles
|
|
Appearance
|
White to Off-white to Beige Crystalline Powder
|
|
Melting Point
|
119-121°C (literature)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong bases and nucleophiles to avoid premature substitution.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Terpyridine ligand synthesis, conjugated polymer precursor, kinase inhibitor scaffold development, agrochemical intermediate, symmetric heterocycle construction, Pd-catalyzed cross-coupling reactions
|
|
Reactivity
|
Suitable for Suzuki coupling, Stille coupling, Negishi coupling, Sonogashira coupling, nucleophilic aromatic substitution (SNAr), and lithiation at C3/C5 positions
|
|
Special Note
|
✅ Symmetric Building Block—C2/C6 dibromo pattern enables streamlined synthesis of C2-symmetric structures. Dual activated sites for sequential functionalization. Part of our halogenated pyridine series.
|