You are here:
- Home
- Aromatic hydrocarbons
- 2-Bromo-6-methoxypyridine
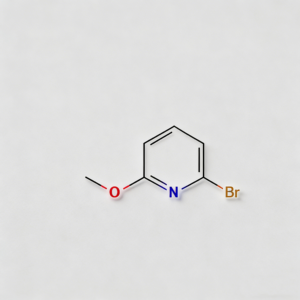
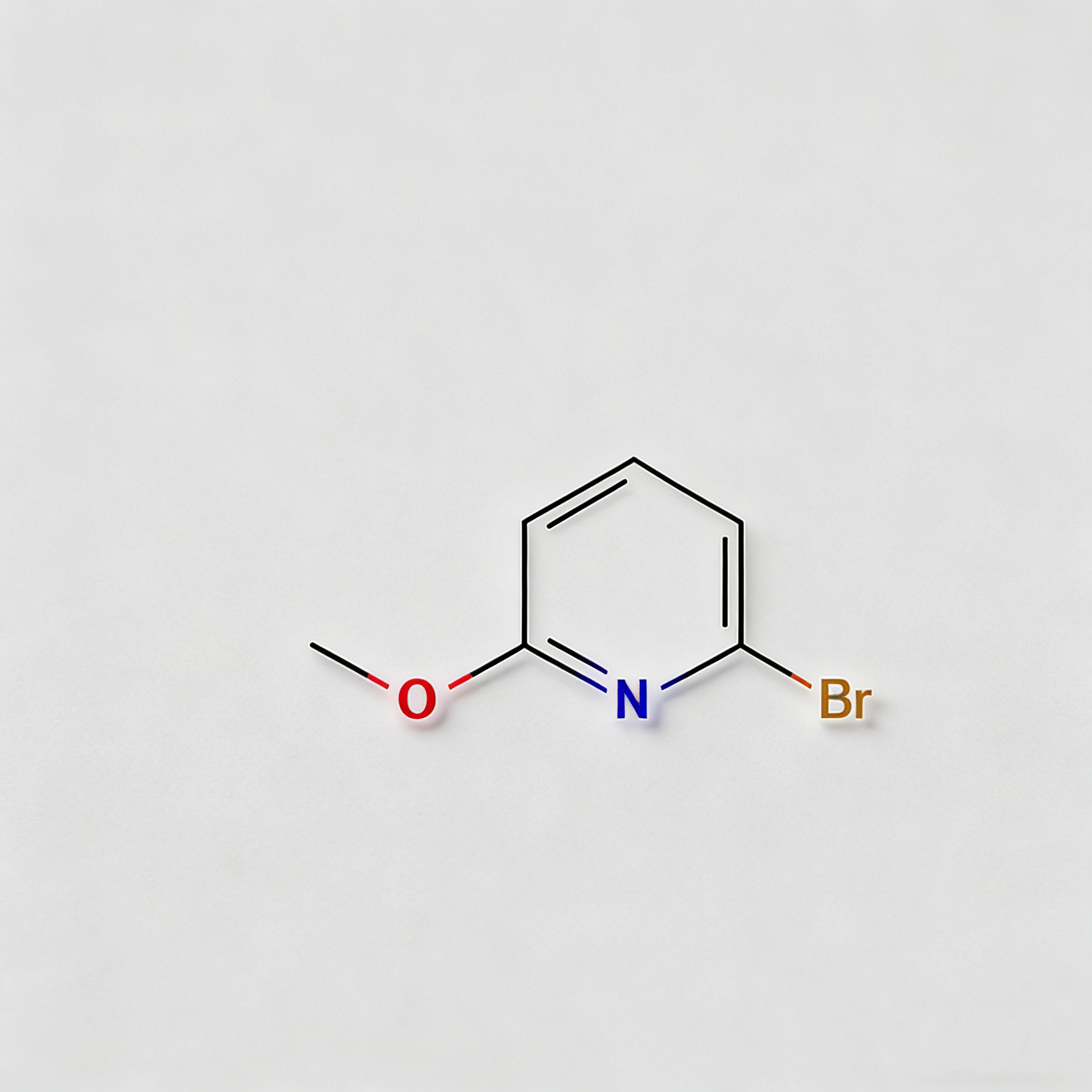
2-Bromo-6-methoxypyridine
2-Bromo-6-methoxypyridine (CAS 40473-07-2) is a strategically asymmetric halogenated heterocyclic scaffold engineered for cross-coupling reactions, ligand synthesis, and pharmaceutical intermediate development. This molecule integrates a pyridine core—a nitrogen-containing six-membered aromatic ring—with a bromine substituent at the C2 position and a methoxy group at the C6 position. The C2-bromo handle is activated by the adjacent ring nitrogen, enabling efficient Pd-catalyzed cross-coupling (Suzuki, Stille, Negishi, Sonogashira) to introduce diverse aryl, alkyl, or heteroaryl groups. The C6-methoxy group serves as an electron-donating substituent that modulates the electronic properties of the pyridine ring, provides steric bulk, and can serve as a protected phenol equivalent (demethylatable to hydroxyl for further functionalization). This 2,6-disubstitution pattern offers a distinct geometry compared to symmetric dibromopyridines, making it particularly valuable for constructing asymmetric ligands, kinase inhibitor cores, and agrochemical actives where specific electronic and steric environments are required. Manufactured under controlled conditions, it supports drug discovery and materials science programs requiring modular access to differentially functionalized pyridine architectures.
Property
|
Product Name
|
2-Bromo-6-methoxypyridine
|
|
CAS Number
|
40473-07-2
|
|
Chinese Name
|
2-溴 -6-甲氧基吡啶
|
|
Synonyms
|
6-Methoxy-2-bromopyridine; Pyridine, 2-bromo-6-methoxy-; C₆H₆BrNO heterocycle; 2-Bromo-6-methoxypyridine
|
|
Chinese Synonyms
|
6-甲氧基 -2-溴吡啶; 溴甲氧基吡啶; 2-溴 -6-甲氧基氮苯; 不对称二取代吡啶
|
|
Molecular Formula
|
C₆H₆BrNO
|
|
Molecular Weight
|
188.02 g/mol
|
|
Purity
|
≥ 98% (GC/HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Pyridine Derivatives / Halogenated Heterocycles
|
|
Appearance
|
Colorless to Pale Yellow Liquid or Low-Melting Solid
|
|
Boiling Point
|
~230-240°C at 760 mmHg (estimated)
|
|
Density
|
~1.55 g/cm³ (estimated)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong acids (to avoid demethylation) and strong bases/nucleophiles (to avoid substitution).
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Kinase inhibitor scaffold development, asymmetric ligand synthesis, agrochemical intermediate, Pd-catalyzed cross-coupling (C2-Br), demethylation to hydroxyl derivative, heterocyclic library construction
|
|
Reactivity
|
Suitable for Suzuki coupling, Stille coupling, Negishi coupling (C2-Br displacement), O-demethylation (e.g., BBr₃), electrophilic aromatic substitution at C3/C4/C5, and directed lithiation
|
|
Special Note
|
✅ Asymmetric 2,6-Disubstituted Scaffold—C2-Br for coupling, C6-OMe for electronic modulation/protection. Distinct from symmetric 2,6-dibromopyridine (CAS 626-05-1) by electronic properties. Part of our halogenated pyridine series.
|