You are here:
- Home
- Aromatic hydrocarbons
- 3,5-Dibromo-2-pyridylamine
3,5-Dibromo-2-pyridylamine
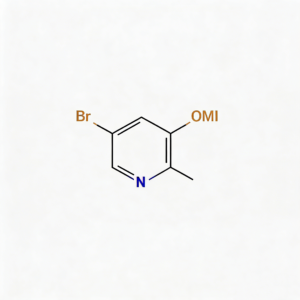
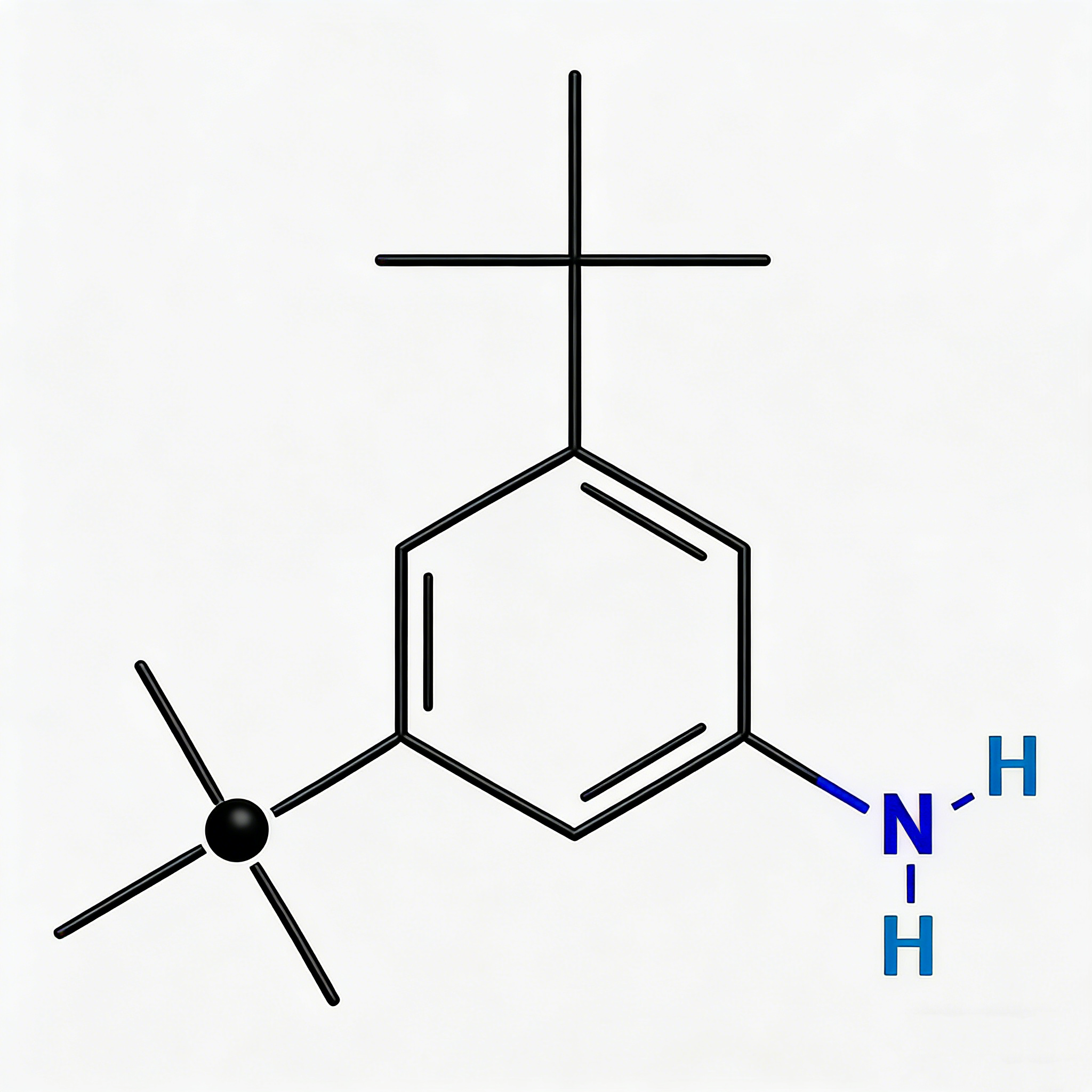
3,5-Dibromo-2-pyridylamine (CAS 35486-42-1), also known as 2-Amino-3,5-dibromopyridine, is a strategically functionalized halogenated heterocyclic scaffold engineered for cross-coupling reactions, pharmaceutical intermediate development, and ligand synthesis. This molecule integrates a pyridine core—a nitrogen-containing six-membered aromatic ring—with an amino substituent at the C2 position and two bromine atoms at the C3 and C5 positions. The C2-amino group provides nucleophilic reactivity for acylation, alkylation, diazotization, or heterocyclic fusion, while the dual bromine handles enable sequential or simultaneous Pd-catalyzed cross-coupling (Suzuki, Stille, Negishi, Sonogashira) to introduce diverse aryl, alkyl, or heteroaryl groups. This tri-functional architecture offers orthogonal reactivity patterns, making it particularly valuable for constructing kinase inhibitor cores, agrochemical actives, and asymmetric ligands where differentiated synthetic strategies are required. Compared to dibromopyridines without amino groups, this compound provides additional derivatization pathways through the nucleophilic nitrogen. Manufactured with purity ≥98% under controlled conditions, it supports drug discovery and materials science programs requiring modular access to differentially functionalized pyridine architectures.
Property
|
Product Name
|
3,5-Dibromo-2-pyridylamine
|
|
CAS Number
|
35486-42-1
|
|
Chinese Name
|
3,5-二溴 -2-吡啶胺
|
|
Synonyms
|
2-Amino-3,5-dibromopyridine; Pyridin-2-amine, 3,5-dibromo-; 2-Pyridylamine, 3,5-dibromo-
|
|
Chinese Synonyms
|
2-氨基 -3,5-二溴吡啶; 3,5-二溴 -2-氨基吡啶; 二溴氨基吡啶; 3,5-二溴氮苯 -2-胺
|
|
Molecular Formula
|
C₅H₄Br₂N₂
|
|
Molecular Weight
|
251.91 g/mol
|
|
Purity
|
≥ 98% (GC/HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Pyridine Derivatives / Halogenated Aminopyridines
|
|
Appearance
|
White to Off-white to Pale Yellow Crystalline Powder
|
|
Melting Point
|
~155 – 157 °C (literature reference recommended)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong oxidizing agents and acids to avoid amino group oxidation or diazotization.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Kinase inhibitor scaffold development, agrochemical intermediate synthesis, Pd-catalyzed cross-coupling (C3/C5-Br), amino group derivatization (acylation/alkylation), heterocyclic library construction, asymmetric ligand synthesis
|
|
Reactivity
|
Suitable for Suzuki coupling, Stille coupling, Negishi coupling (C3/C5-Br displacement), amino group acylation/alkylation, diazotization, heterocyclic fusion, and electrophilic aromatic substitution at C4/C6
|
|
Special Note
|
✅ Tri-Functional Scaffold—C2-NH₂ for nucleophilic derivatization, C3/C5-Br for cross-coupling. Orthogonal Reactivity—amino and bromo groups can be independently modified. Distinct from dibromopyridines by amino functionality. Part of our halogenated aminopyridine series.
|