You are here:
- Home
- Aromatic hydrocarbons
- 3-(4-Chlorobenzoyl)propionic acid
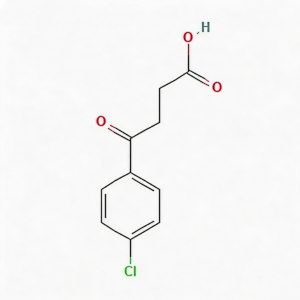
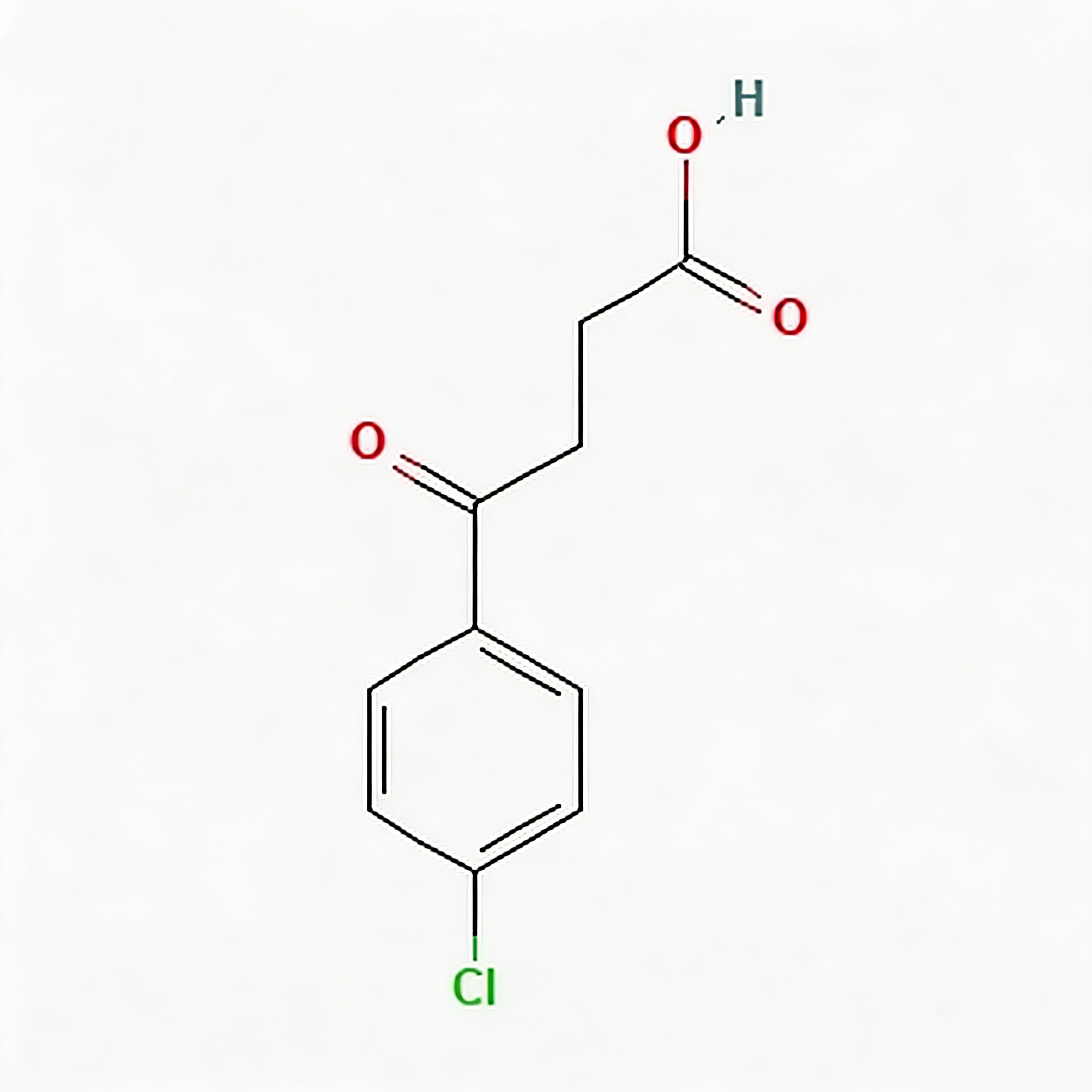
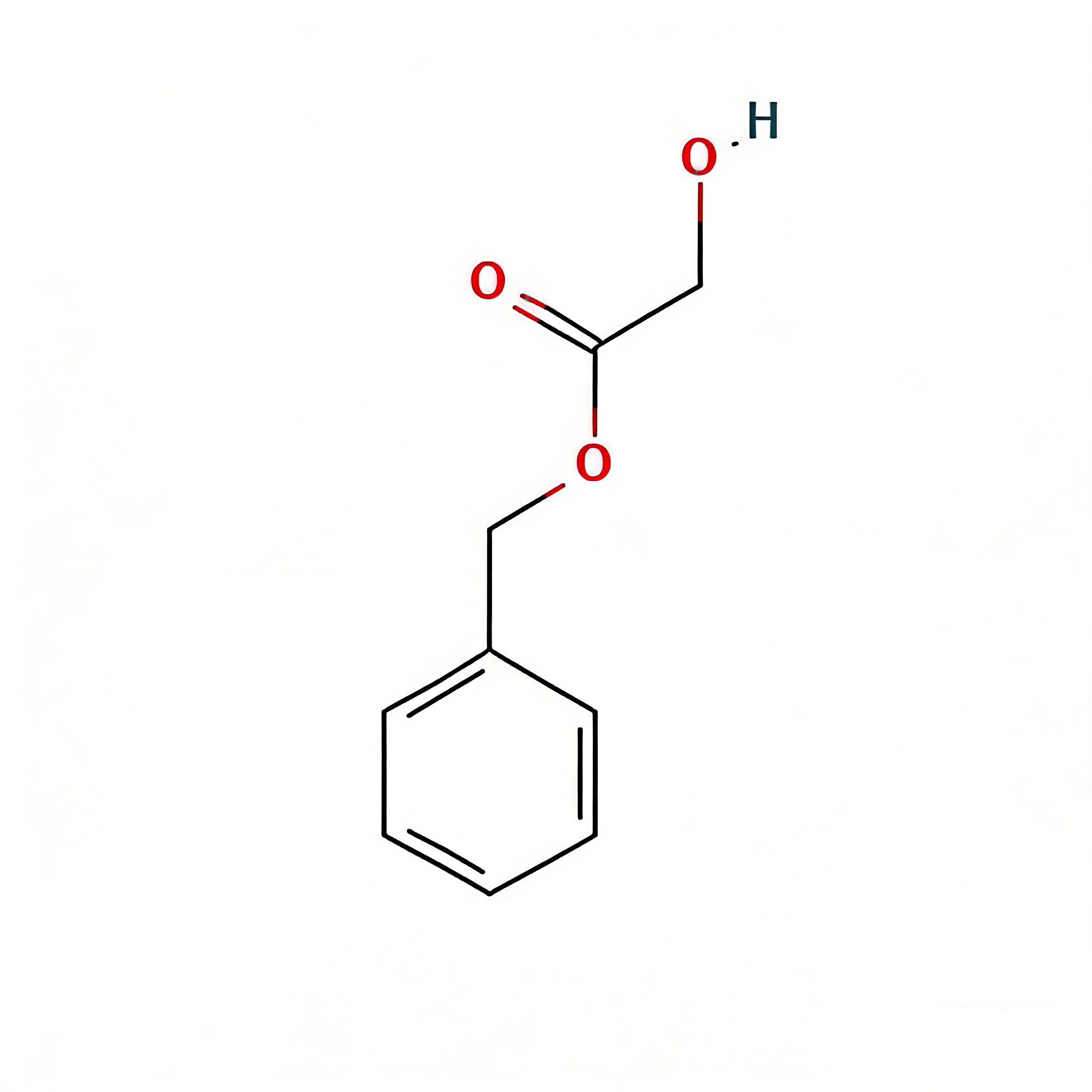
3-(4-Chlorobenzoyl)propionic acid
3-(4-Chlorobenzoyl)propionic acid (CAS 3984-34-7) is a strategically functionalized γ-keto acid engineered for heterocyclic construction, pharmaceutical intermediate synthesis, and agrochemical development. This molecule integrates a 4-chlorophenyl core—a halogenated aromatic system with enhanced metabolic stability—with a benzoyl ketone linker and a terminal propionic acid moiety. The γ-keto acid architecture (Ar-CO-CH₂-CH₂-COOH) serves as a privileged scaffold for Paal-Knorr pyrrole synthesis, Friedländer quinoline construction, and Knoevenagel condensation reactions. The 4-chloro substituent provides an additional handle for late-stage functionalization via cross-coupling or nucleophilic aromatic substitution, while the carboxylic acid terminus enables salt formation, amide coupling, or esterification for formulation optimization. This intermediate is particularly valuable for constructing NSAID analogs, PPAR modulators, antifungal agents, and herbicide scaffolds where the chlorobenzoyl-propionic acid motif balances lipophilicity with versatile synthetic reactivity. Manufactured with purity ≥98% under controlled conditions, it supports drug discovery programs requiring modular access to halogenated keto-acid architectures with defined substitution patterns.
Property
|
Product Name
|
3-(4-Chlorobenzoyl)propionic acid
|
|
CAS Number
|
3984-34-7
|
|
Chinese Name
|
3-(4-氯苯甲酰基) 丙酸
|
|
Synonyms
|
4-Chlorobenzoylpropionic acid; 4-(4-Chlorophenyl)-4-oxobutanoic acid; γ-(4-Chlorobenzoyl)butyric acid; C₁₀H₉ClO₃ keto-acid
|
|
Chinese Synonyms
|
4-氯苯甲酰丙酸; 4-(4-氯苯基)-4-氧代丁酸; γ-(4-氯苯甲酰) 丁酸; 氯代酮酸中间体
|
|
Molecular Formula
|
C₁₀H₉ClO₃
|
|
Molecular Weight
|
212.63 g/mol
|
|
Purity
|
≥ 98% (HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Halogenated Aromatic Acids / Keto-Acid Derivatives
|
|
Appearance
|
White to orange to green crystalline powder
|
|
Melting Point
|
~ 142-146°C (literature reference recommended)
|
|
pKa
|
~4.39 ± 0.10 (carboxylic acid, estimated)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed. Protect from strong bases to avoid decarboxylation or Claisen condensation side reactions.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100g/bottle, 500g/bottle, 1kg/bottle, 5kg/drum, or customized according to customer requirements
|
|
Applications
|
Heterocyclic synthesis (pyrroles/quinolines via Paal-Knorr/Friedländer), NSAID analog development, PPAR modulator research, agrochemical intermediate synthesis, Knoevenagel condensation precursor, amide coupling for drug conjugates
|
|
Reactivity
|
Suitable for Paal-Knorr pyrrole synthesis, Friedländer quinoline construction, Knoevenagel condensation, amide coupling (activation via EDC/HATU), esterification, decarboxylative coupling, and electrophilic aromatic substitution on the chlorophenyl ring
|