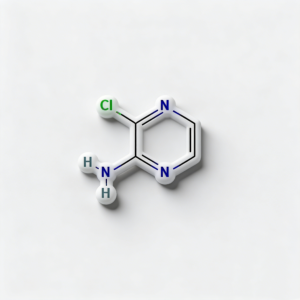
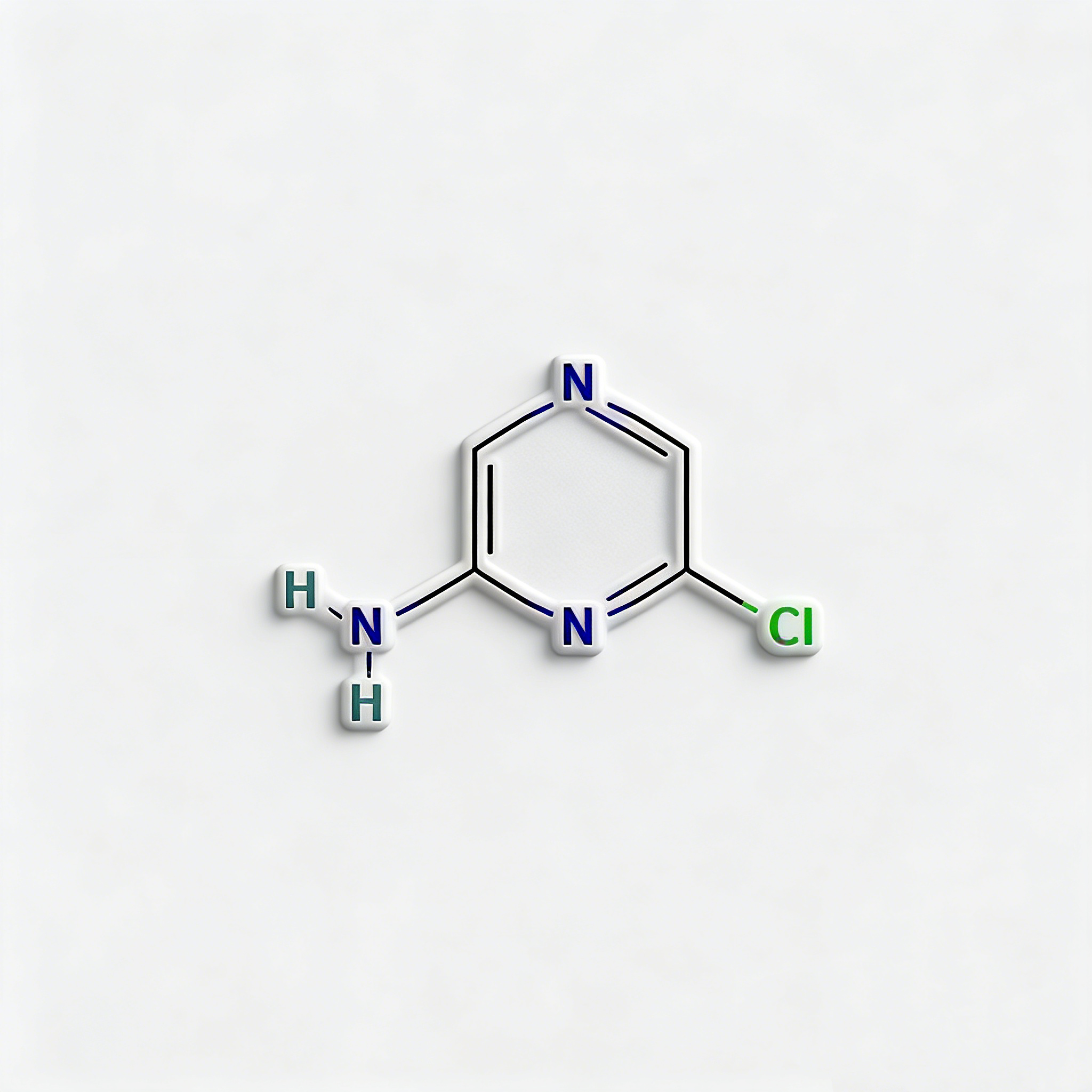
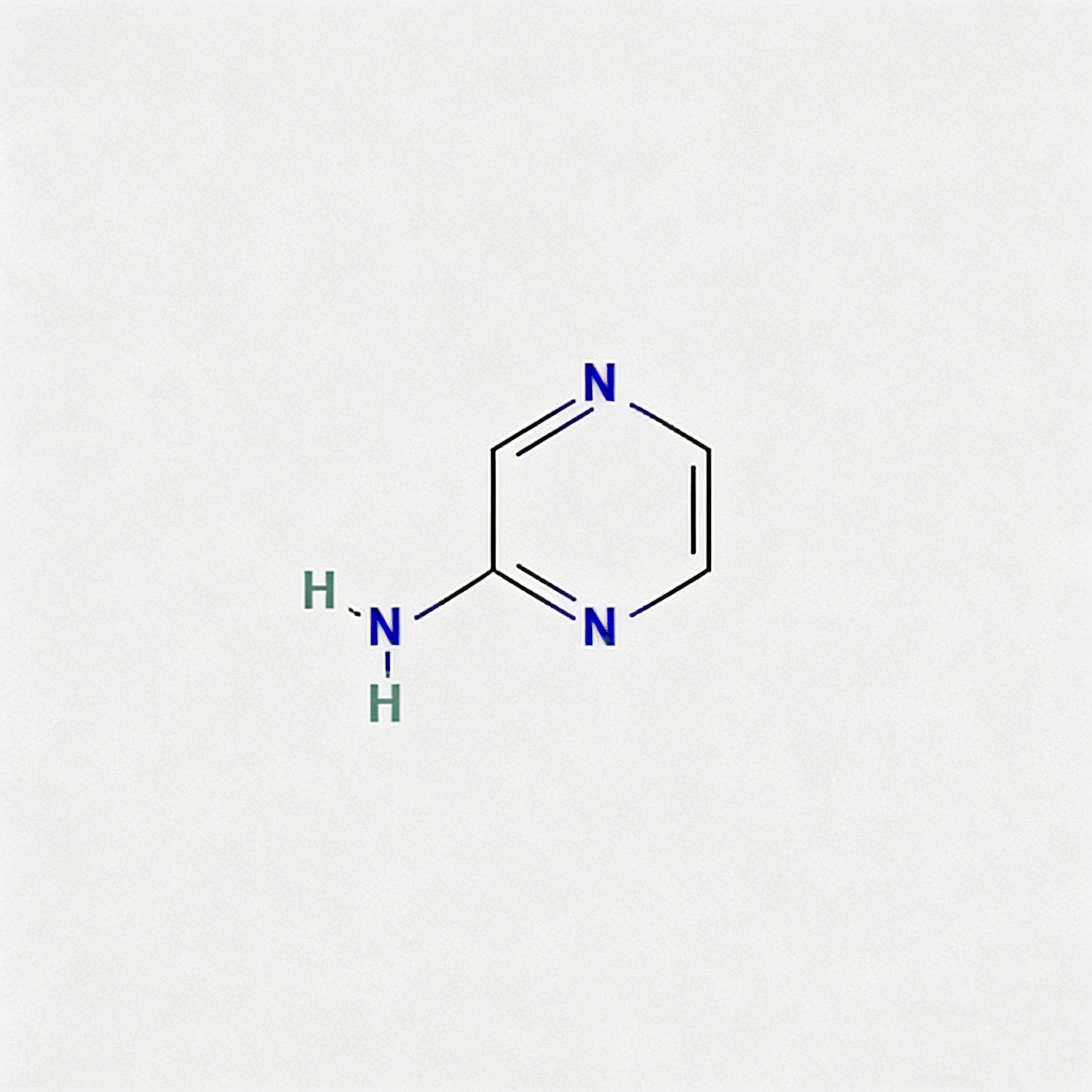
2-Amino-3-chloropyrazine
2-Amino-3-chloropyrazine (CAS 6863-73-6) is a strategically bifunctional pyrazine scaffold engineered for heterocyclic diversification, pharmaceutical intermediate synthesis, and agrochemical development. This molecule integrates an electron-deficient pyrazine core—a six-membered aromatic diazine ring with nitrogen atoms at para positions (1,4)—with an amino substituent at the C2 position and a chlorine atom at the C3 position. The adjacent 2,3-substitution pattern creates unique electronic and steric properties compared to the 2,6-isomer (CAS 33332-28-4), enabling distinct reactivity profiles for sequential functionalization. The C3-chloro handle is activated by the adjacent ring nitrogen and amino group, enabling efficient nucleophilic aromatic substitution (SNAr) or Pd-catalyzed cross-coupling, while the C2-amino group provides orthogonal reactivity for acylation, alkylation, diazotization, or heterocyclic fusion. This dual-functionality architecture is particularly valuable for constructing fused heterocycles (pyrazino-fused systems), kinase inhibitor cores, and nitrogen-rich ligands where the ortho-chloroamino-pyrazine motif enables unique cyclization pathways and binding geometries. Manufactured with purity ≥98% under controlled conditions, it supports drug discovery programs requiring modular access to differentially functionalized electron-deficient heterocycles with vicinal substitution patterns.
Property
|
Product Name
|
2-Amino-3-chloropyrazine
|
|
CAS Number
|
6863-73-6
|
|
Chinese Name
|
2-氨基 -3-氯吡嗪
|
|
Synonyms
|
3-Chloro-2-aminopyrazine; 3-Chloropyrazin-2-amine; Pyrazine, 3-chloro-2-amino-; C₄H₄ClN₃ heterocycle
|
|
Chinese Synonyms
|
3-氯 -2-氨基吡嗪; 3-氯吡嗪 -2-胺; 氯氨基吡嗪 (2,3-位); 邻氯氨基吡嗪
|
|
Molecular Formula
|
C₄H₄ClN₃
|
|
Molecular Weight
|
129.55 g/mol
|
|
Purity
|
≥ 98% (HPLC/GC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Pyrazine Derivatives / Halogenated Aminoheterocycles
|
|
Appearance
|
White to pale yellow to dark green crystalline powder
|
|
Melting Point
|
~ 152-156°C (decomposition, literature reference recommended)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong oxidizing agents to avoid amino group oxidation and from strong bases to avoid premature chloride displacement.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Kinase inhibitor synthesis, antiviral drug development, agrochemical intermediate synthesis, heterocyclic library construction (pyrazino-fused systems via ortho-cyclization), sequential functionalization via orthogonal Cl/NH₂ reactivity, ligand synthesis for coordination chemistry
|
|
Reactivity
|
Suitable for nucleophilic aromatic substitution (C3-Cl displacement), amino group acylation/alkylation, diazotization, heterocyclic fusion (e.g., pyrazino[2,3-d]pyrimidine via ortho-cyclization), electrophilic aromatic substitution at C5/C6, and Pd-catalyzed cross-coupling
|
|
Special Note
|
✅ Ortho-Bifunctional Scaffold—2,3-adjacent Cl/NH₂ enables unique cyclization pathways vs. 2,6-isomer (CAS 33332-28-4). Electron-deficient pyrazine core provides unique binding geometry. Part of our halogenated aminopyrazine series.
|