You are here:
- Home
- Aromatic hydrocarbons
- 2-BROMOPHENETHYLALCOHOL
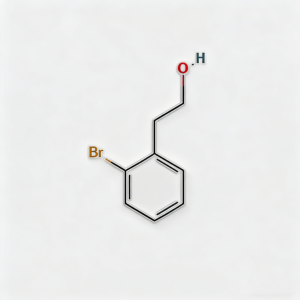
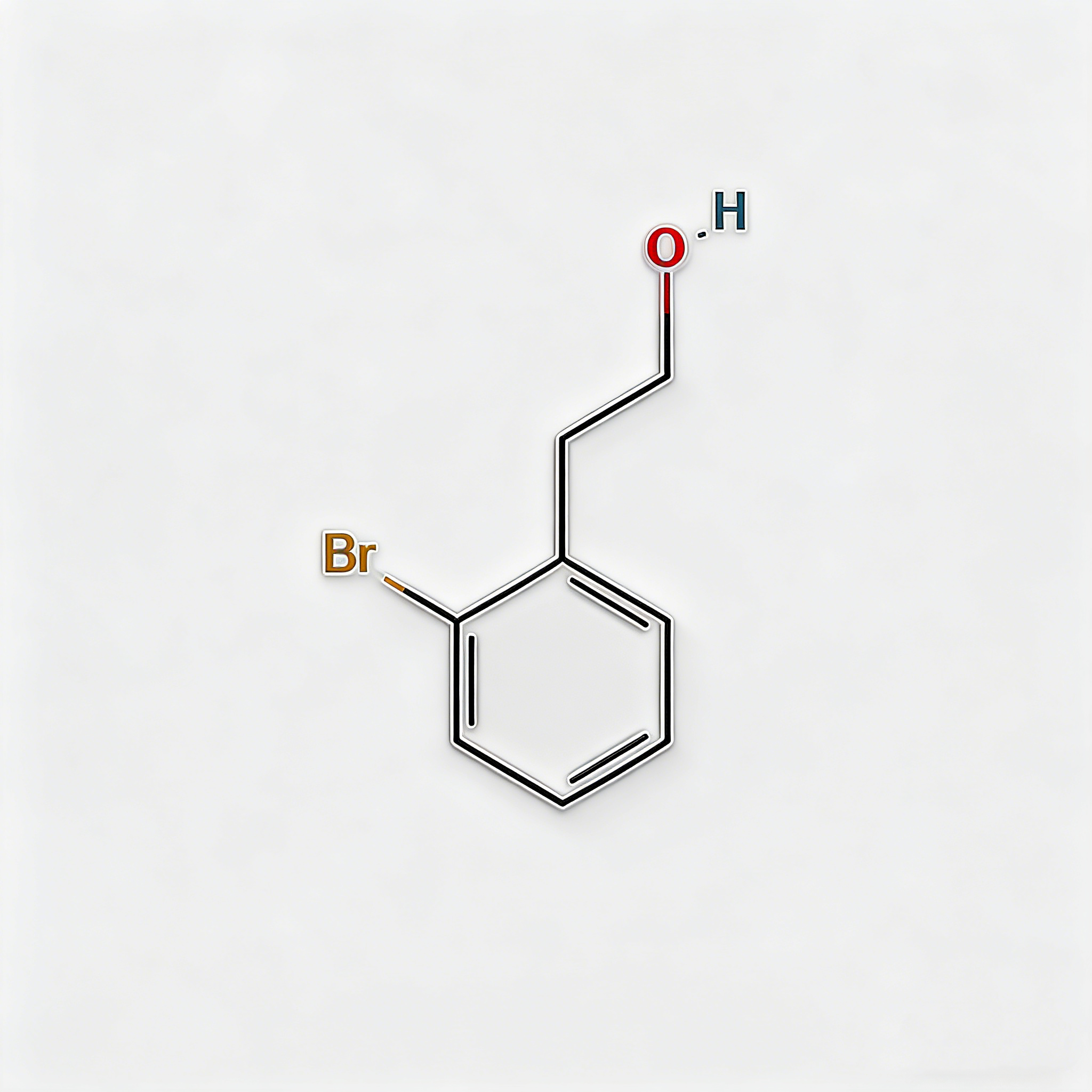
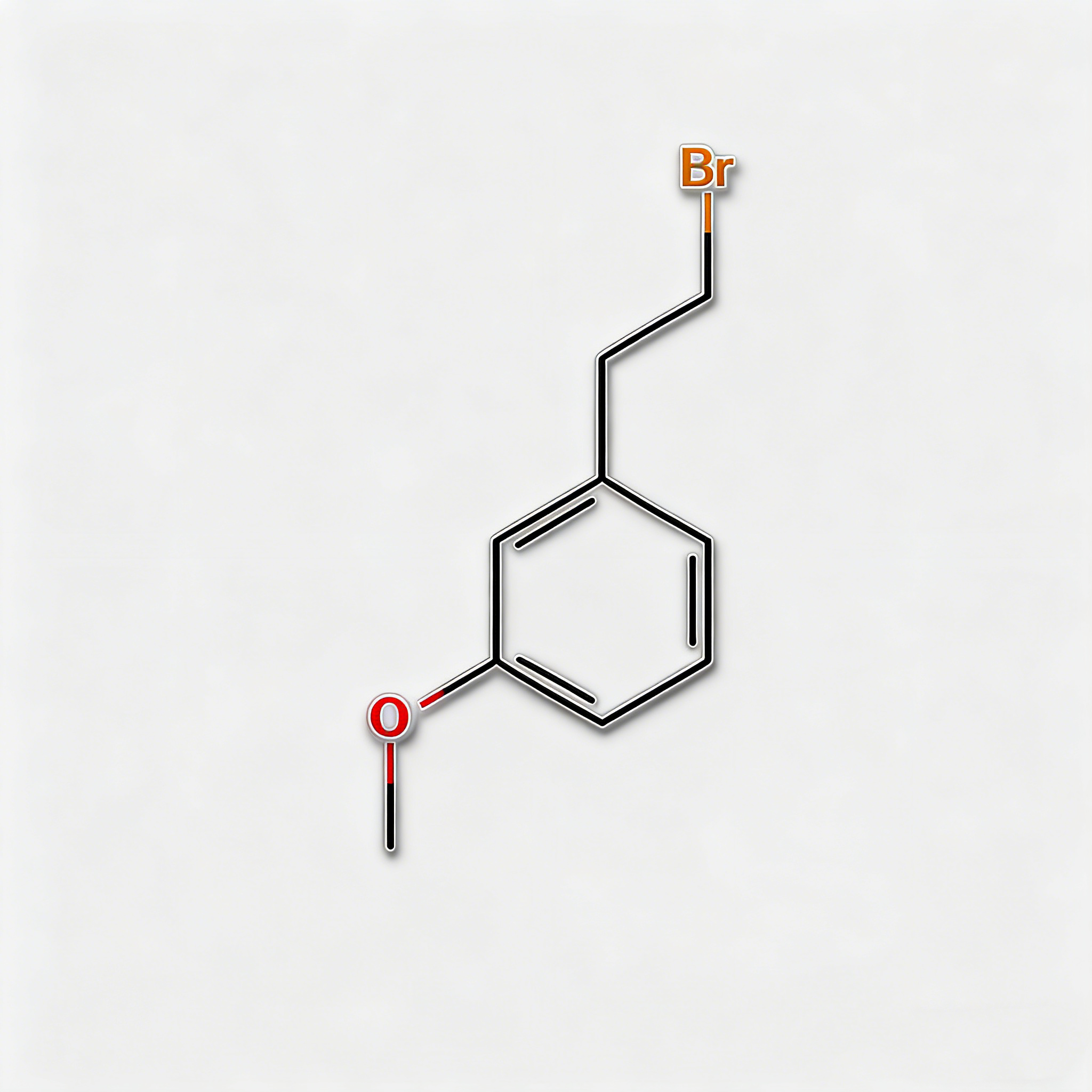
2-BROMOPHENETHYLALCOHOL
2-(2-Bromophenyl)ethanol (CAS 1074-16-4), also known as o-Bromophenethyl alcohol, is a high-purity brominated aromatic alcohol intermediate widely used in pharmaceutical synthesis and organic chemistry research. This compound features a bromine substituent at the ortho-position of the phenethyl alcohol structure, making it a versatile building block for the development of complex organic molecules and active pharmaceutical ingredients (APIs). As a key intermediate in cross-coupling reactions, nucleophilic substitution, and functional group transformations, it exhibits excellent reactivity in Suzuki, Heck, and Sonogashira coupling reactions. Our product is manufactured under strict quality control standards, ensuring a purity of ≥98%, making it ideal for pharmaceutical manufacturing, medicinal chemistry research, and scale-up production. This compound is particularly valuable in the synthesis of pharmaceuticals containing brominated aromatic structures and serves as an important precursor in the preparation of fine chemicals, agrochemical intermediates, and specialty materials.
Property
|
Product Name
|
2-(2-Bromophenyl)ethanol
|
|
CAS Number
|
1074-16-4
|
|
Chinese Name
|
邻溴苯乙醇
|
|
Synonyms
|
o-Bromophenethyl alcohol; 2-Bromophenethyl alcohol; 2-(o-Bromophenyl)ethanol; Benzeneethanol, 2-bromo-; 2-Bromo-phenethyl alcohol; 1-(2-Bromophenyl)-2-ethanol; o-Bromophenylethanol
|
|
Chinese Synonyms
|
2-溴苯乙醇; 邻溴苯基乙醇; 2-(2-溴苯基)乙醇
|
|
Molecular Formula
|
C₈H₉BrO
|
|
Molecular Weight
|
201.06 g/mol
|
|
Purity
|
≥ 98%
|
|
Product Category
|
Pharmaceutical Intermediates / Benzene Derivatives / Phenethyl Alcohols
|
|
Appearance
|
Colorless to Light Yellow Liquid
|
|
Boiling Point
|
~260°C (estimated)
|
|
Density
|
~1.5 g/cm³ (estimated)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
25kg/drum, 50kg/drum, or customized according to customer requirements
|
|
Applications
|
Pharmaceutical intermediates, organic synthesis, API manufacturing, cross-coupling reactions, nucleophilic substitution, fine chemical production
|
|
Reactivity
|
Suitable for Suzuki coupling, Heck reaction, Sonogashira coupling, oxidation to aldehyde/acid, esterification, and etherification reactions
|