You are here:
- Home
- Aromatic hydrocarbons
- 2-Hydroxy-5-bromopyridine
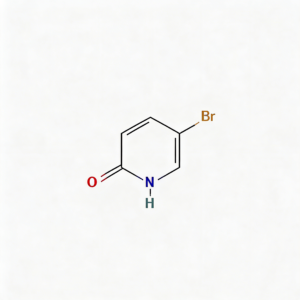
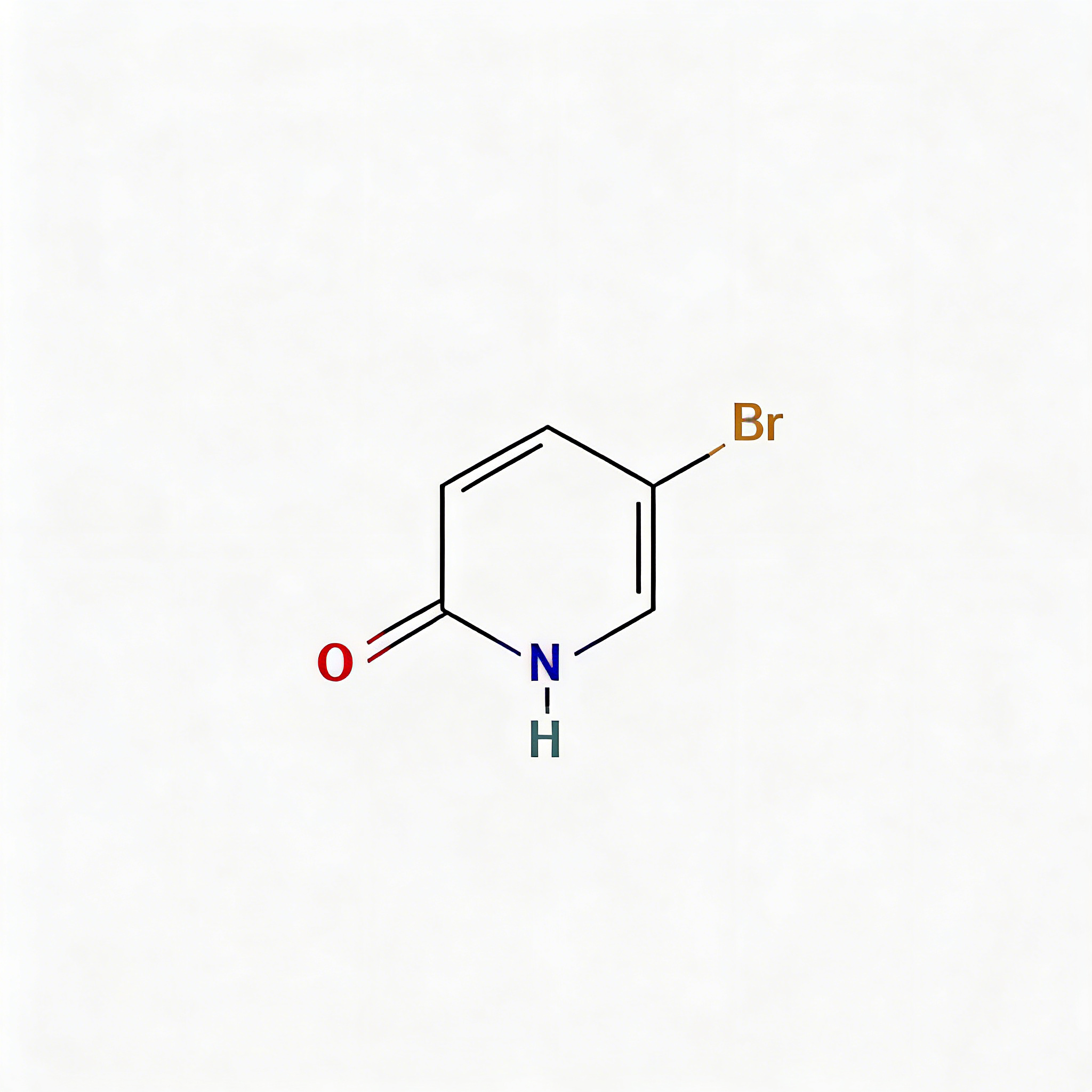
2-Hydroxy-5-bromopyridine
2-Hydroxy-5-bromopyridine (CAS 13466-38-1) is a strategically functionalized pyridine scaffold engineered for pharmaceutical intermediate synthesis, cross-coupling reactions, and heterocyclic diversification . This molecule exists in tautomeric equilibrium between the 2-hydroxypyridine and 2-pyridone forms, integrating a bromine substituent at the C5 position of the pyridine ring. The C5-bromo handle is activated for Pd-catalyzed cross-coupling (Suzuki, Buchwald-Hartwig, Sonogashira) while the 2-hydroxy/oxo group enables hydrogen-bonding interactions and serves as a handle for etherification or alkylation . The compound features an electron-rich C-5 position that enables selective copper-catalyzed C–N bond formation with amines, heterocycles, and amides under mild conditions using inexpensive 1,2-diol ligands such as ethylene glycol . This intermediate is particularly valuable for constructing pharmaceutical actives, agrochemical ingredients, and functional materials where the 2-hydroxy-5-bromo motif enables modular synthetic strategies. Manufactured under controlled conditions, it supports drug discovery and chemical development programs requiring access to functionalized pyridine architectures
Property
|
Product Name
|
2-Hydroxy-5-bromopyridine
|
|
CAS Number
|
13466-38-1
|
|
Chinese Name
|
2-羟基 -5-溴吡啶
|
|
Synonyms
|
5-Bromo-2-pyridinol; 5-Bromo-2-hydroxypyridine; 5-Bromopyridin-2-ol;
|
|
Chinese Synonyms
|
5-溴 -2-吡啶醇; 5-溴 -2-羟基吡啶; 溴羟基吡啶; 2-羟基 -5-溴氮苯
|
|
Molecular Formula
|
C₅H₄BrNO
|
|
Molecular Weight
|
174g/mol
|
|
Purity
|
≥ 98% (HPLC/GC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Pyridine Derivatives / Halogenated Hydroxypyridines
|
|
Appearance
|
White to Off-white Crystalline Powder or Solid
|
|
Melting Point
|
~180-185°C (literature reference recommended; may exist as pyridone tautomer)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong oxidizing agents and bases to avoid oxidation or substitution.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Kinase inhibitor scaffold development, agrochemical intermediate synthesis, Pd-catalyzed cross-coupling (C5-Br), triflate formation (C2-OH), heterocyclic library construction, functional material precursor
|
|
Reactivity
|
Suitable for Suzuki coupling, Stille coupling, Negishi coupling (C5-Br displacement), triflation (C2-OH to OTf), O-alkylation, tautomerization control, and electrophilic aromatic substitution at C3/C4/C6
|
|
Special Note
|
✅ Dual-Functionality Scaffold—C5-Br for coupling, C2-OH for triflation/alkylation. Tautomeric Equilibrium—exists as 2-hydroxypyridine/2-pyridone forms. Distinct from dibromopyridines by OH/Br differentiation. Part of our halogenated pyridine series.
|