You are here:
- Home
- Pyrimidines
- 3-Bromo-4-methyl-5-nitropyridine
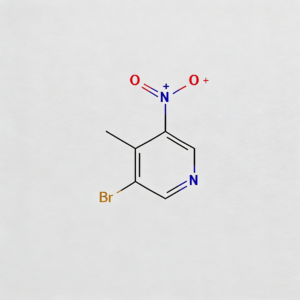
3-Bromo-4-methyl-5-nitropyridine
3-Bromo-4-methyl-5-nitropyridine (CAS 69872-15-7) is a strategically tri-substituted pyridine scaffold engineered for regioselective diversification in medicinal chemistry and agrochemical R&D. Featuring bromine at C3, methyl at C4, and nitro at C5, this polyfunctionalized heterocycle enables orthogonal synthetic manipulation: the C3-bromo substituent serves as a privileged handle for Pd-catalyzed cross-coupling (Suzuki, Buchwald-Hartwig, Sonogashira), while the C5-nitro group offers a reducible motif to access amino-pyridine pharmacophores, and the C4-methyl group provides steric and electronic tuning for target binding optimization. This intermediate is particularly valuable for constructing kinase inhibitor cores, neonicotinoid analogs, and heterocyclic APIs where precise 3,4,5-trisubstitution patterns modulate biological activity. Manufactured with purity ≥98% under stringent QC protocols, it supports scalable routes to complex pyridine-based therapeutics requiring meta/para-disubstituted aromatic architectures.
Property
|
Product Name
|
3-Bromo-4-methyl-5-nitropyridine
|
|
CAS Number
|
69872-15-7
|
|
Chinese Name
|
3-溴-4-甲基-5-硝基吡啶
|
|
Synonyms
|
3-Bromo-4-methyl-5-nitro-1-azabenzene; Pyridine, 3-bromo-4-methyl-5-nitro-; 3-Bromo-5-nitro-4-picoline; C₆H₅BrN₂O₂ heterocycle; MFCD11847767
|
|
Chinese Synonyms
|
3-溴-4-甲基-5-硝基-1-氮杂苯; 3-溴-5-硝基-4-甲基吡啶; 溴甲基硝基吡啶; 3-溴-4-甲基-5-硝基吡啶
|
|
Molecular Formula
|
C₆H₅BrN₂O₂
|
|
Molecular Weight
|
217.02 g/mol
|
|
Purity
|
≥ 98% (GC/HPLC)
|
|
Product Category
|
Pharmaceutical Intermediates / Pyridine Derivatives / Polyfunctionalized Nitropyridines
|
|
Appearance
|
Yellow to Brown Crystalline Powder or Solid
|
|
Melting Point
|
57-58°C (literature)
|
|
Boiling Point
|
~320°C at 760 mmHg (estimated)
|
|
Density
|
~1.75 g/cm³ (estimated)
|
|
Flash Point
|
116.3±25.9°C
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong reducing agents to prevent premature nitro reduction.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100g/bottle, 500g/bottle, 1kg/bottle, 5kg/drum, or customized according to customer requirements
|
|
Applications
|
Kinase inhibitor synthesis, neonicotinoid agrochemical development, regioselective pyridine functionalization, nitro reduction to amino, C3-selective cross-coupling, methyl group C–H functionalization strategies
|
|
Reactivity
|
Suitable for Suzuki coupling, Buchwald-Hartwig amination, Sonogashira coupling (preferential C3-Br displacement), catalytic hydrogenation (nitro to amino), electrophilic aromatic substitution at C6, and methyl group oxidation/functionalization
|
|
Special Note
|
✅ Light-sensitive solid with orthogonal reactivity—C3-Br for coupling, C5-NO₂ for reduction, C4-Me for steric modulation. Part of our pyridine derivative series.
|