You are here:
- Home
- Aromatic hydrocarbons
- 3-Bromoquinoline
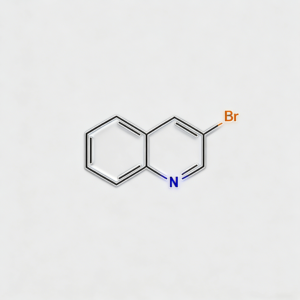
3-Bromoquinoline
3-Bromoquinoline (CAS 5332-24-1) is a strategically functionalized fused bicyclic heterocyclic scaffold engineered for cross-coupling reactions, pharmaceutical intermediate development, and materials science applications. This molecule integrates a quinoline core—a fused benzene-pyridine bicyclic system prevalent in bioactive natural products and pharmaceuticals (including antimalarial drugs like quinine and chloroquine)—with a bromine substituent at the C3 position. The C3-bromo handle serves as a privileged site for Pd-catalyzed cross-coupling (Suzuki, Stille, Negishi, Sonogashira), enabling efficient introduction of diverse aryl, alkyl, or heteroaryl groups to the quinoline scaffold. The fused bicyclic architecture provides enhanced rigidity, extended π-conjugation, and improved metabolic stability compared to monocyclic pyridine analogs, making it particularly valuable for constructing kinase inhibitor cores, antimalarial agents, antimicrobial compounds, and functional materials where the quinoline motif offers superior target binding affinity and photophysical properties. Compared to halogenated pyridines, quinoline derivatives provide expanded aromatic surface area for π-stacking interactions and distinct electronic distribution across the fused ring system. Manufactured with purity ≥98% under controlled conditions, it supports drug discovery and materials science programs requiring modular access to functionalized quinoline architectures.
Property
|
Product Name
|
3-Bromoquinoline
|
|
CAS Number
|
5332-24-1
|
|
Chinese Name
|
3-溴喹啉
|
|
Synonyms
|
Quinoline, 3-bromo-; 3-Bromoquinoline; C₉H₆BrN heterocycle; 3-Bromo-1-azanaphthalene
|
|
Chinese Synonyms
|
3-溴 -1-氮杂萘; 溴喹啉; 3-位溴代喹啉; 喹啉 -3-溴
|
|
Molecular Formula
|
C₉H₆BrN
|
|
Molecular Weight
|
208.05 g/mol
|
|
Purity
|
≥ 98% (GC/HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Quinoline Derivatives / Halogenated Heterocycles
|
|
Appearance
|
Clear colorless to yellow liquid
|
|
Melting Point
|
~26-30°C (literature reference recommended)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong oxidizing agents and bases to avoid oxidation or substitution.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Kinase inhibitor scaffold development, antimalarial drug synthesis (chloroquine analogs), agrochemical intermediate, Pd-catalyzed cross-coupling (C3-Br), functional material precursor, heterocyclic library construction
|
|
Reactivity
|
Suitable for Suzuki coupling, Stille coupling, Negishi coupling, Sonogashira coupling (C3-Br displacement), electrophilic aromatic substitution at C5/C6/C7/C8, and directed lithiation
|
|
Special Note
|
✅ Fused Bicyclic Scaffold—extended π-system vs. monocyclic pyridines. C3-Br offers strategic functionalization site. Distinct from pyridine derivatives by benzene fusion. Part of our halogenated quinoline series.
|