You are here:
- Home
- Aromatic hydrocarbons
- 4-bromo-2-hydroxyacetophenone
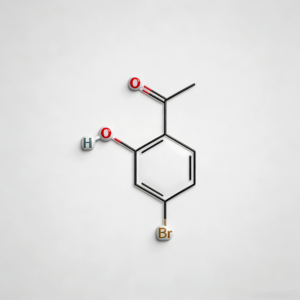
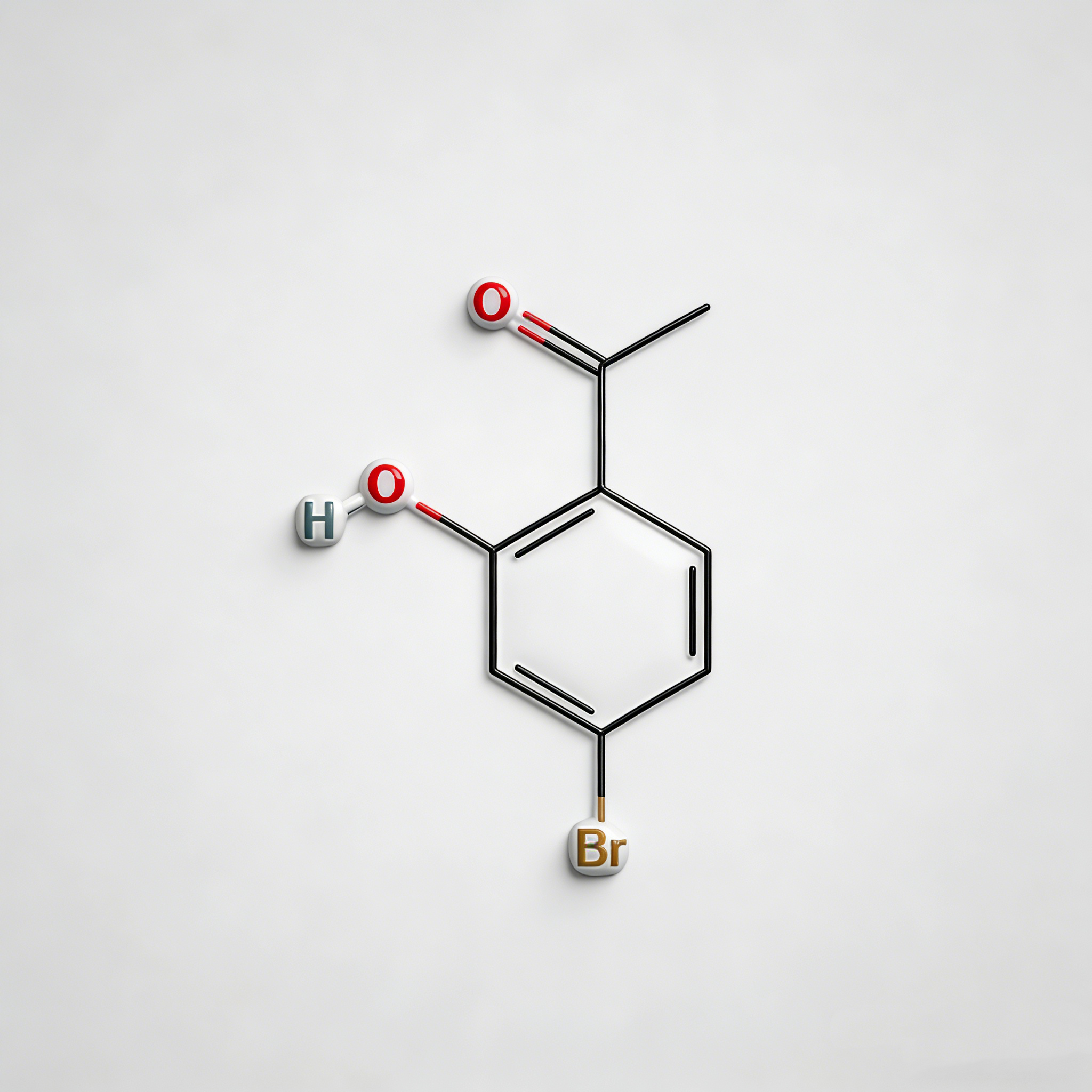
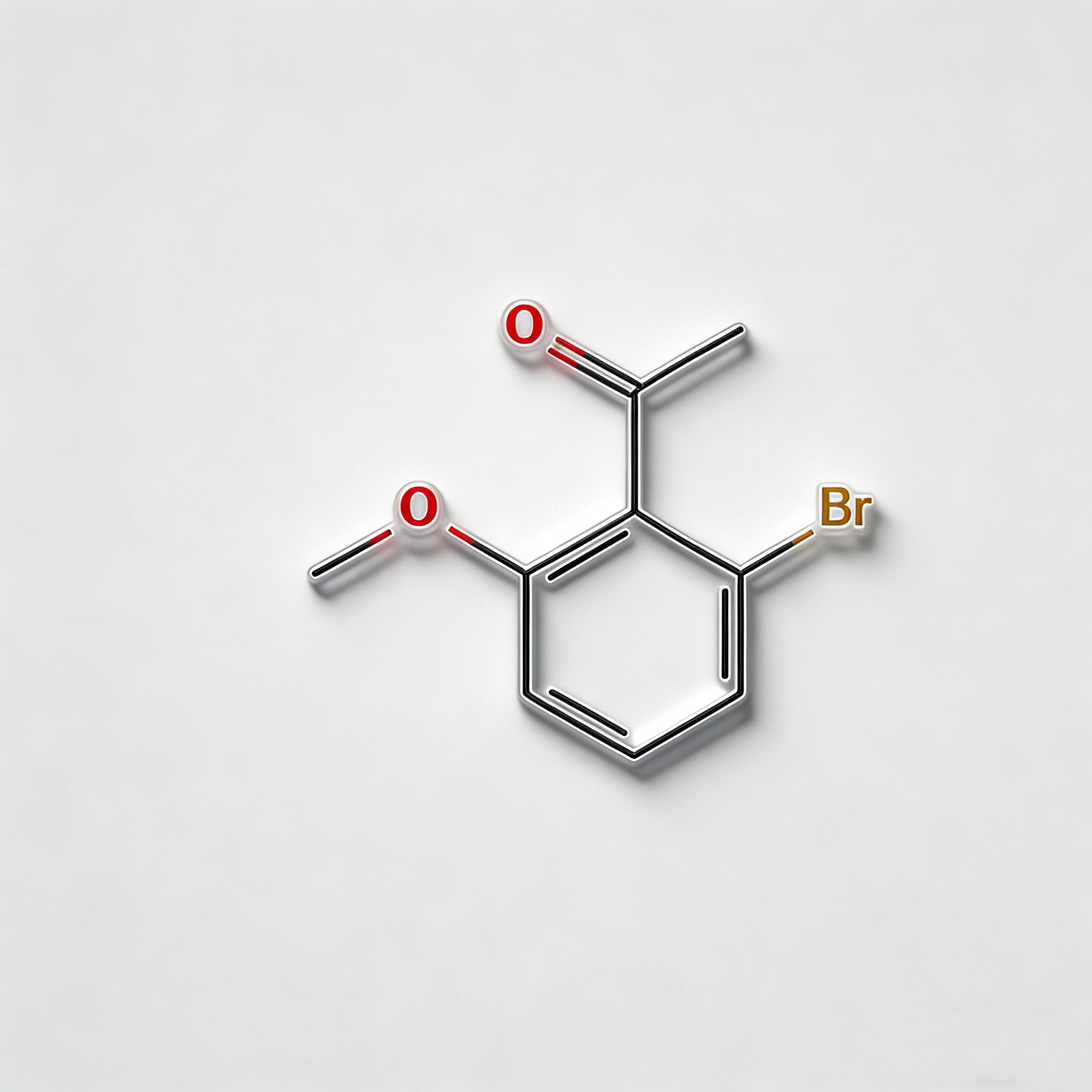
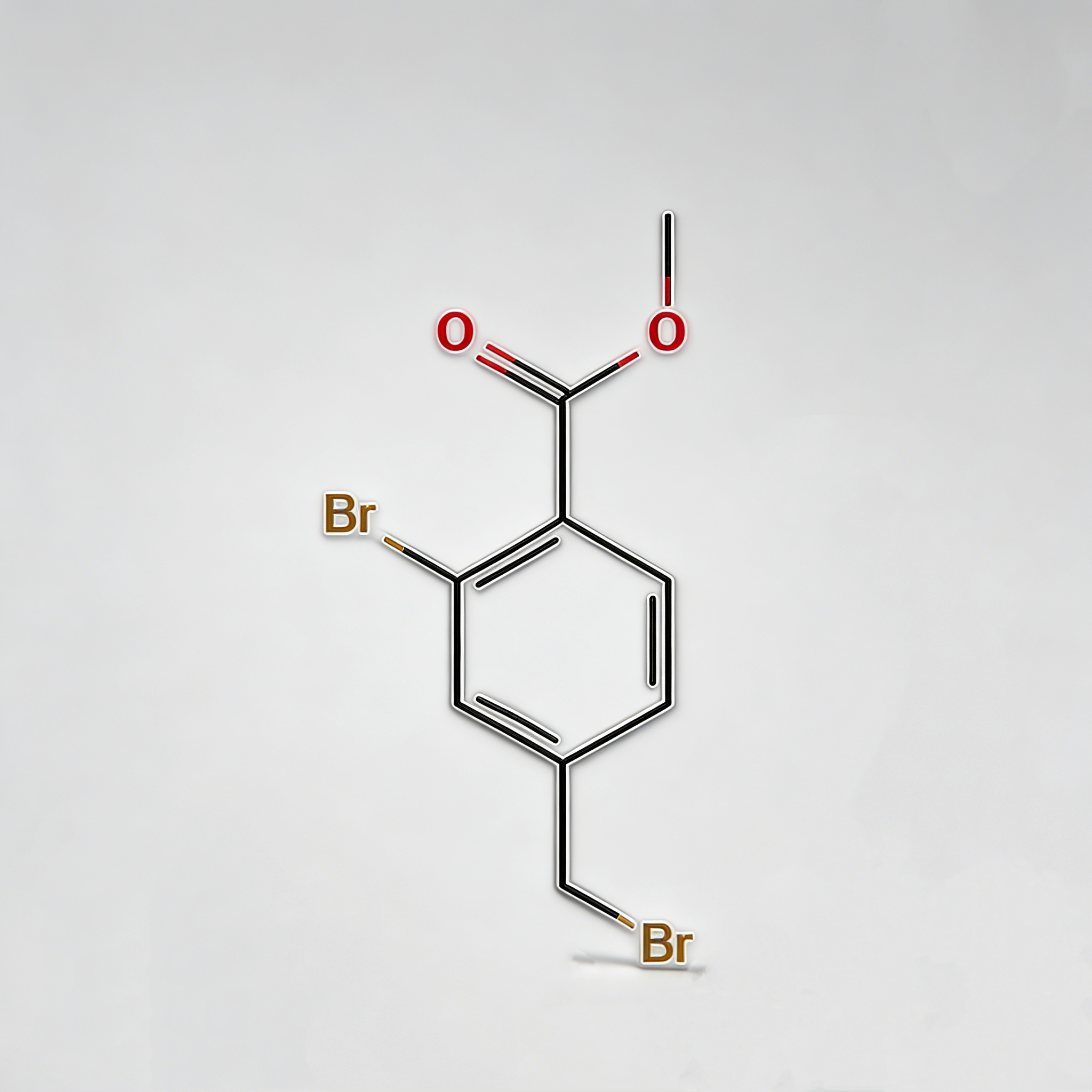
4-bromo-2-hydroxyacetophenone
4-Bromo-2-hydroxyacetophenone (CAS 30186-18-6) is a high-purity brominated hydroxyacetophenone intermediate widely used in pharmaceutical synthesis and organic chemistry research. This compound features a bromine substituent at the para-position and a hydroxyl group at the ortho-position of the acetophenone structure, making it a versatile building block for the development of complex organic molecules and active pharmaceutical ingredients (APIs). As a key intermediate in heterocyclic synthesis, condensation reactions, and functional group transformations, it exhibits excellent reactivity in Friedel-Crafts acylation, Claisen-Schmidt condensation, and cyclization reactions. Our product is manufactured under strict quality control standards, ensuring a purity of ≥98%, making it ideal for pharmaceutical manufacturing, medicinal chemistry research, and scale-up production. This compound is particularly valuable in the synthesis of pharmaceuticals containing brominated phenolic structures, benzofuran derivatives, and chromone-based APIs.
Property
|
Product Name
|
4-Bromo-2-hydroxyacetophenone
|
|
CAS Number
|
30186-18-6
|
|
Chinese Name
|
4-溴-2-羟基苯乙酮
|
|
Synonyms
|
1-(4-Bromo-2-hydroxyphenyl)ethanone; 2′-Hydroxy-4′-bromoacetophenone; Ethanone, 1-(4-bromo-2-hydroxyphenyl)-; 4-Bromosalicyloyl methyl ketone; o-Hydroxy-p-bromoacetophenone; 4-Bromo-2-hydroxyphenyl methyl ketone; 2-Hydroxy-4-bromoacetophenone
|
|
Chinese Synonyms
|
2-羟基-4-溴苯乙酮; 1-(4-溴-2-羟基苯基)乙酮; 4-溴邻羟基苯乙酮; 邻羟基对溴苯乙酮
|
|
Molecular Formula
|
C₈H₇BrO₂
|
|
Molecular Weight
|
215.04 g/mol
|
|
Purity
|
≥ 98%
|
|
Product Category
|
Pharmaceutical Intermediates / Acetophenone Derivatives / Brominated Phenols
|
|
Appearance
|
White to Off-white Crystalline Powder
|
|
Melting Point
|
135-138°C (literature)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed. Protect from oxidation.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
1kg/foil bag, 5kg/drum, 25kg/drum, or customized according to customer requirements
|
|
Applications
|
Pharmaceutical intermediates, organic synthesis, API manufacturing, heterocyclic synthesis, benzofuran/chromone preparation, condensation reactions
|
|
Reactivity
|
Suitable for Claisen-Schmidt condensation, cyclization reactions, etherification, esterification, and nucleophilic substitution reactions
|
|
Special Note
|
Key intermediate for benzofuran and chromone scaffold synthesis
|