You are here:
- Home
- Aromatic hydrocarbons
- 5-Chloro-2,4-difluorobenzenesulfonyl chloride
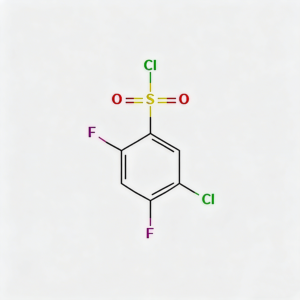
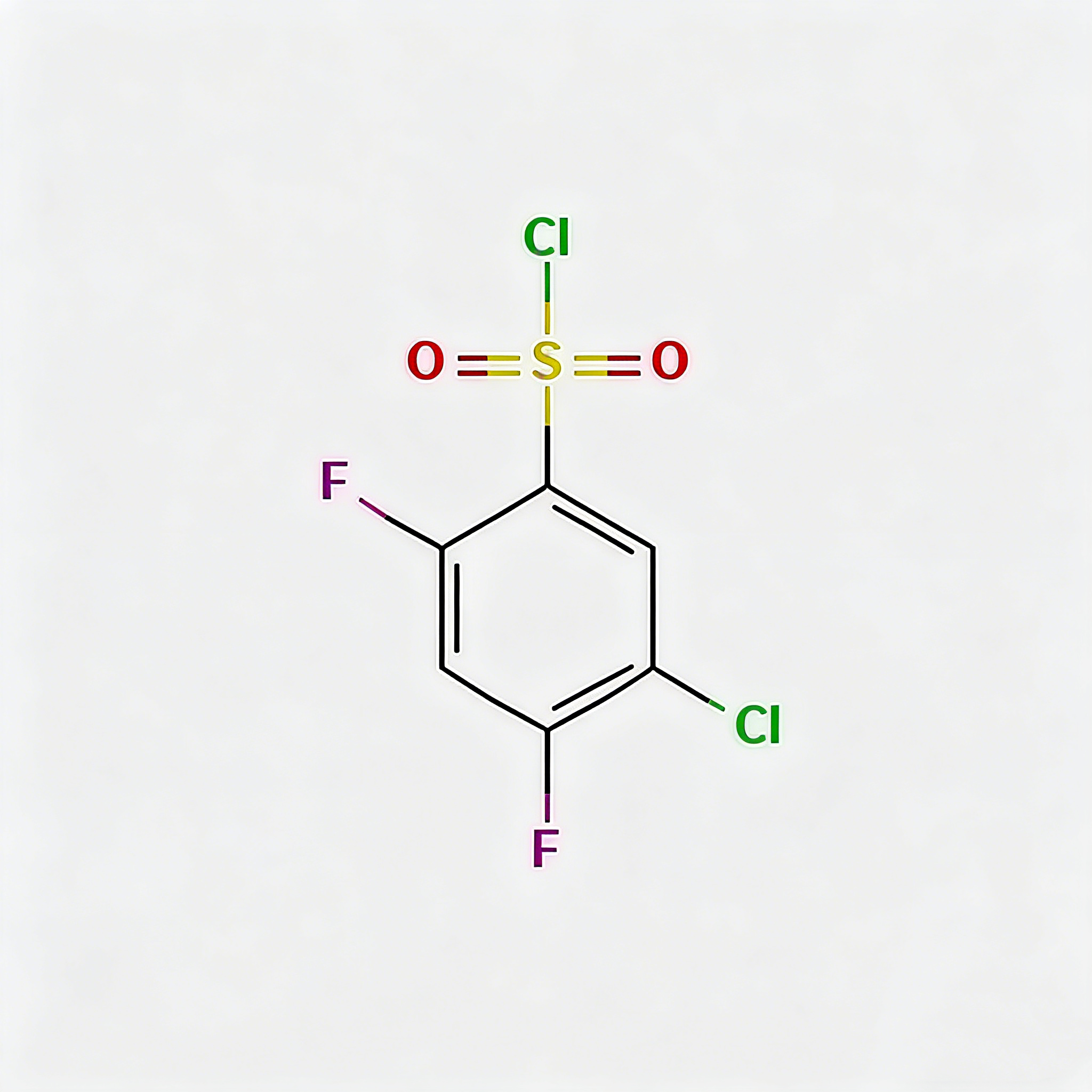
5-Chloro-2,4-difluorobenzenesulfonyl chloride
5-Chloro-2,4-difluorobenzenesulfonyl chloride (CAS 13656-57-0) is a high-purity fluorinated benzene sulfonyl chloride intermediate widely used in pharmaceutical synthesis and organic chemistry research. This compound features both chloro and difluoro substituents on the benzene ring, making it a highly reactive electrophilic reagent for sulfonylation reactions. As a key building block in the synthesis of pharmaceuticals, agrochemicals, and specialty chemicals, it exhibits excellent reactivity in nucleophilic substitution reactions. Our product is manufactured under strict quality control standards, ensuring a purity of ≥98%, making it ideal for API synthesis, medicinal chemistry research, and industrial applications. This compound is particularly valuable in the development of sulfonamide-based drugs and fluorinated pharmaceutical intermediates.
Property
|
Product Name
|
5-Chloro-2,4-difluorobenzenesulfonyl chloride
|
|
CAS Number
|
13656-57-0
|
|
Chinese Name
|
5-氯-2,4-二氟苯磺酰氯
|
|
Synonyms
|
5-Chloro-2,4-difluorobenzene-1-sulfonyl chloride; Benzenesulfonyl chloride, 5-chloro-2,4-difluoro-; 5-Chloro-2,4-difluorobenzenesulfonyl chloride; CDFSC; 2,4-Difluoro-5-chlorobenzenesulfonyl chloride
|
|
Chinese Synonyms
|
5-氯-2,4-二氟苯磺酸氯; 5-氯-2,4-二氟苯-1-磺酰氯
|
|
Molecular Formula
|
C₆H₂Cl₂F₂O₂S
|
|
Molecular Weight
|
243.05 g/mol
|
|
Purity
|
≥ 98%
|
|
Product Category
|
Pharmaceutical Intermediates / Benzene Derivatives / Sulfonyl Chlorides
|
|
Appearance
|
White to off-white crystalline solid |
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from moisture and light. Keep container tightly closed. Moisture sensitive.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
25kg/drum, 50kg/drum, or customized according to customer requirements
|
|
Applications
|
Pharmaceutical intermediates, sulfonylation reactions, API synthesis, agrochemical intermediates, sulfonamide drug development
|
|
Reactivity
|
Suitable for sulfonylation reactions, nucleophilic substitution, sulfonamide synthesis
|