You are here:
- Home
- Aromatic hydrocarbons
- Methyl 4-bromo-2-bromomethylbenzoate
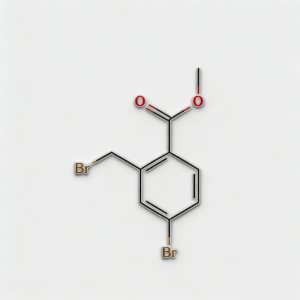
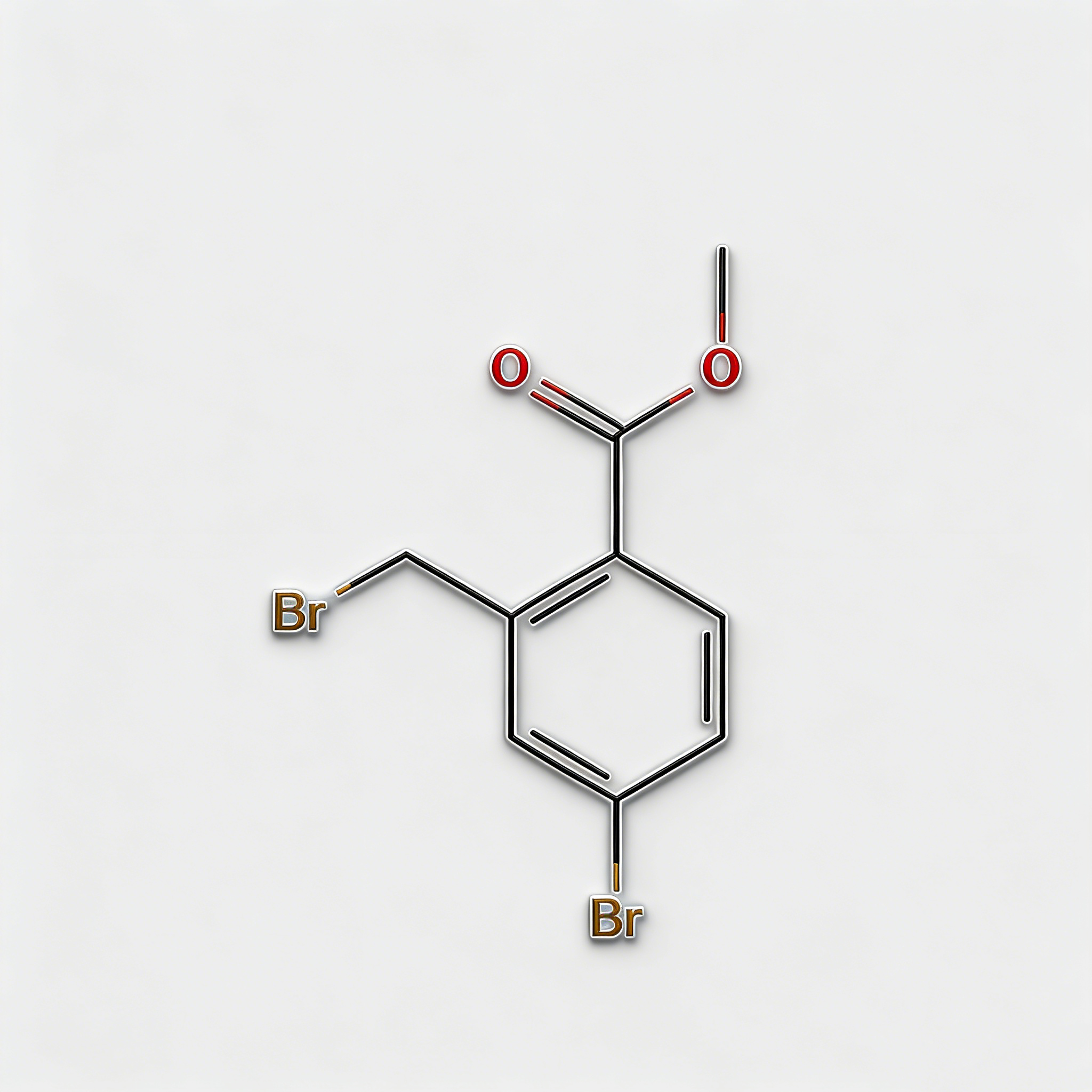
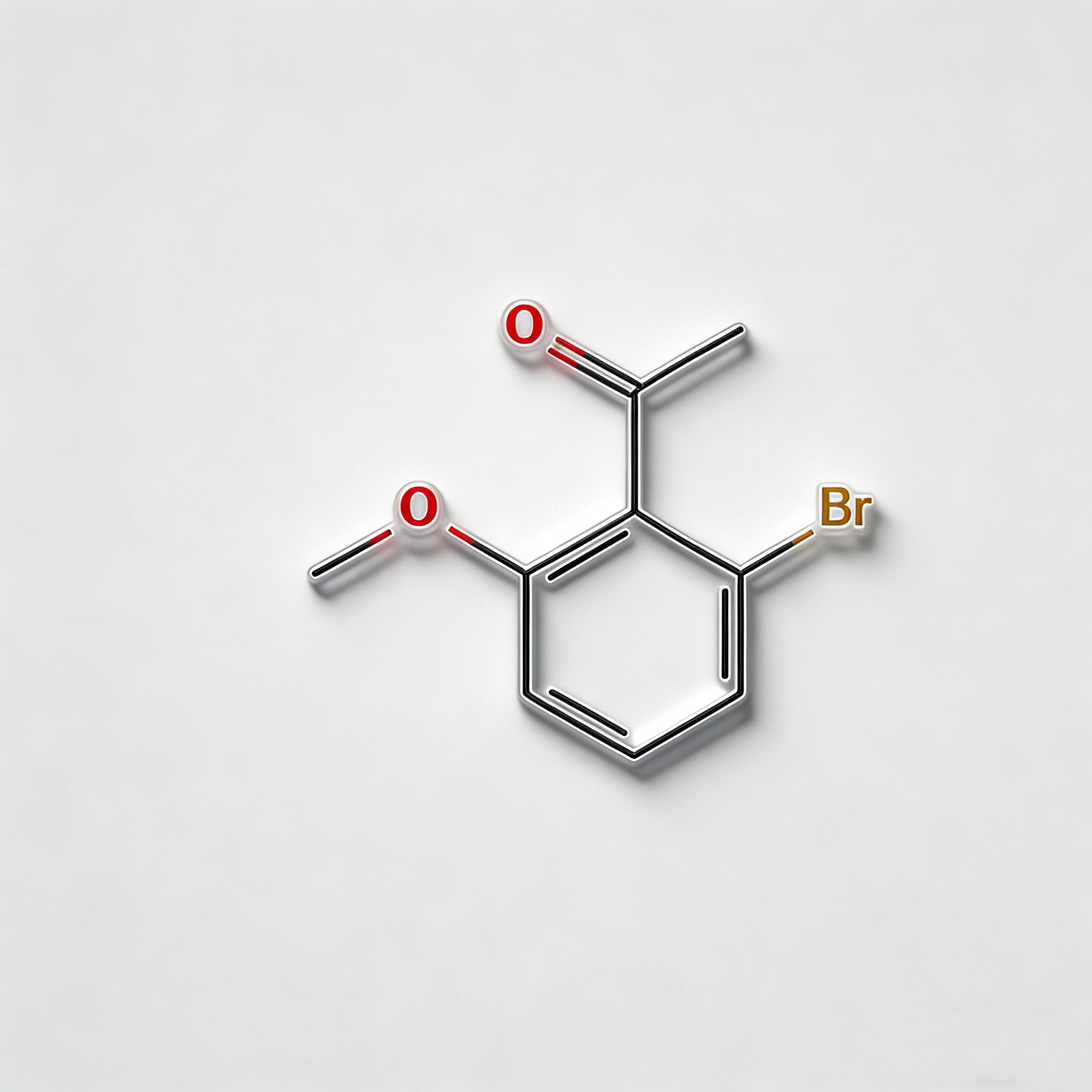
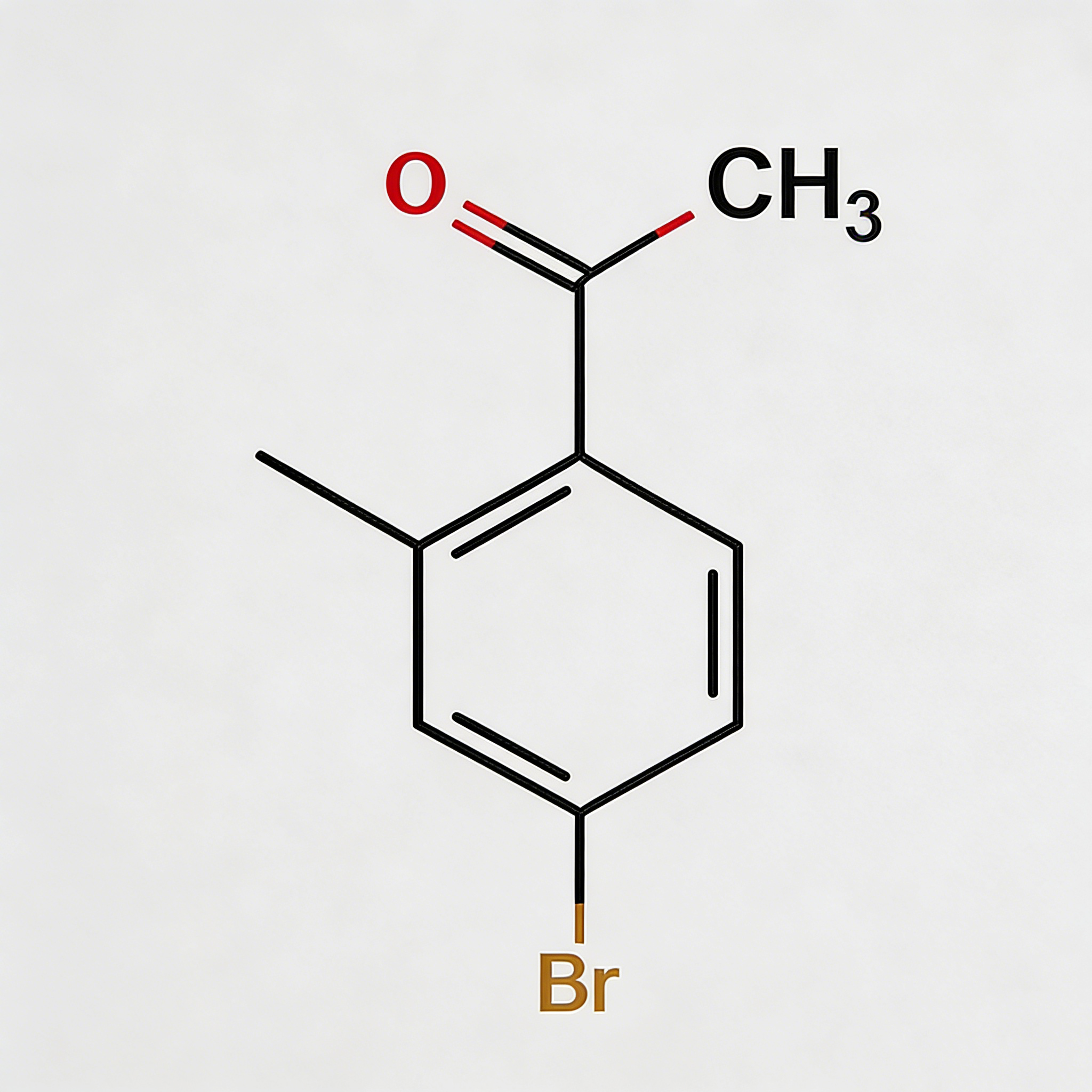
Methyl 4-bromo-2-bromomethylbenzoate
Methyl 4-bromo-2-bromomethylbenzoate (CAS 78471-43-9) is a strategically bifunctional halogenated aromatic ester engineered for pharmaceutical intermediate synthesis, agrochemical development, and materials science applications. This molecule integrates a benzoate core—a benzene ring with a methyl ester substituent—with two distinct bromine functionalities: an aryl bromine at the C4 position and a benzyl bromine (bromomethyl) at the C2 position. This dual-bromine architecture with differentiated reactivity enables orthogonal synthetic strategies: the benzyl bromide is highly reactive towards nucleophilic substitution (SN2), while the aryl bromide is suited for Pd-catalyzed cross-coupling (Suzuki, Stille, Negishi). The methyl ester moiety provides additional versatility for hydrolysis to the acid, transesterification, or amide coupling. Compared to its acid analog (CAS 68837-59-2), this ester form offers enhanced lipophilicity and compatibility with base-sensitive reactions. This intermediate is particularly valuable for constructing complex biaryl systems, fused heterocycles, kinase inhibitor cores, and functional materials where sequential functionalization at the bromine sites is critical. Manufactured with purity ≥98% under controlled conditions, it supports drug discovery programs requiring modular access to differentially halogenated aromatic esters with tunable reactivity.
Property
|
Product Name
|
Methyl 4-bromo-2-bromomethylbenzoate
|
|
CAS Number
|
78471-43-9
|
|
Chinese Name
|
4-溴-2-溴甲基苯甲酸甲酯
|
|
Synonyms
|
Methyl 2-(bromomethyl)-4-bromobenzoate; Benzoic acid, 4-bromo-2-(bromomethyl)-, methyl ester;
|
|
Chinese Synonyms
|
4-溴-2-溴甲基苯甲酸甲酯; 2-溴甲基-4-溴苯甲酸甲酯
|
|
Molecular Formula
|
C₉H₈Br₂O₂
|
|
Molecular Weight
|
307.97 g/mol
|
|
Purity
|
≥ 98% (HPLC/GC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Benzoic Acid Derivatives / Orthogonal Dihalides
|
|
Appearance
|
White to Off-white Crystalline Solid or Low-melting Solid
|
|
Melting Point
|
~50-55°C (estimated, may exist as low-melting solid)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong bases to avoid ester hydrolysis or benzyl bromide substitution. Handle with care (lachrymator/alkylating agent).
|
|
Shelf Life
|
18-24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements (amber glass recommended)
|
|
Applications
|
Kinase inhibitor synthesis, biaryl construction via sequential coupling, heterocyclic library construction, agrochemical intermediate synthesis, functional material precursor, orthogonal functionalization studies
|
|
Reactivity
|
Suitable for nucleophilic substitution (C2-CH₂Br), Pd-catalyzed cross-coupling (C4-ArBr), ester hydrolysis, transesterification, amide coupling (after hydrolysis), and sequential derivatization
|
|
Special Note
|
✅ Orthogonal Dihalo Scaffold—Benzyl Br (SN2) vs. Aryl Br (Cross-coupling). Potent Alkylating Agent—handle with appropriate PPE (gloves, goggles, fume hood). Distinct from mono-bromo analogs by dual reactivity. Part of our halogenated aromatic ester series.
|