You are here:
- Home
- Aromatic hydrocarbons
- tert-Butyl 4-bromo-2-methylbenzoate
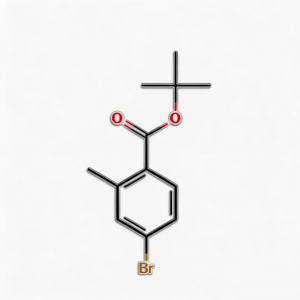
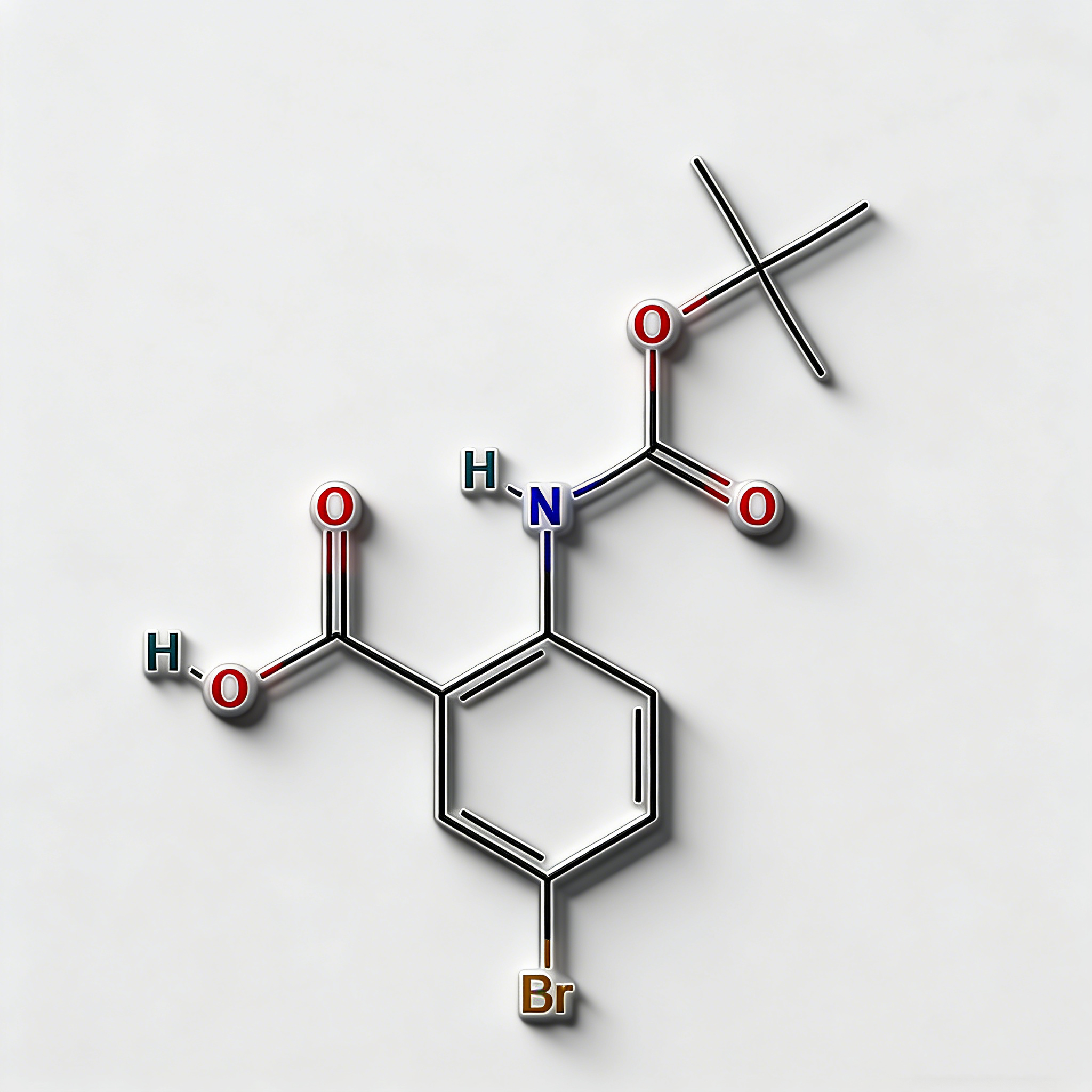
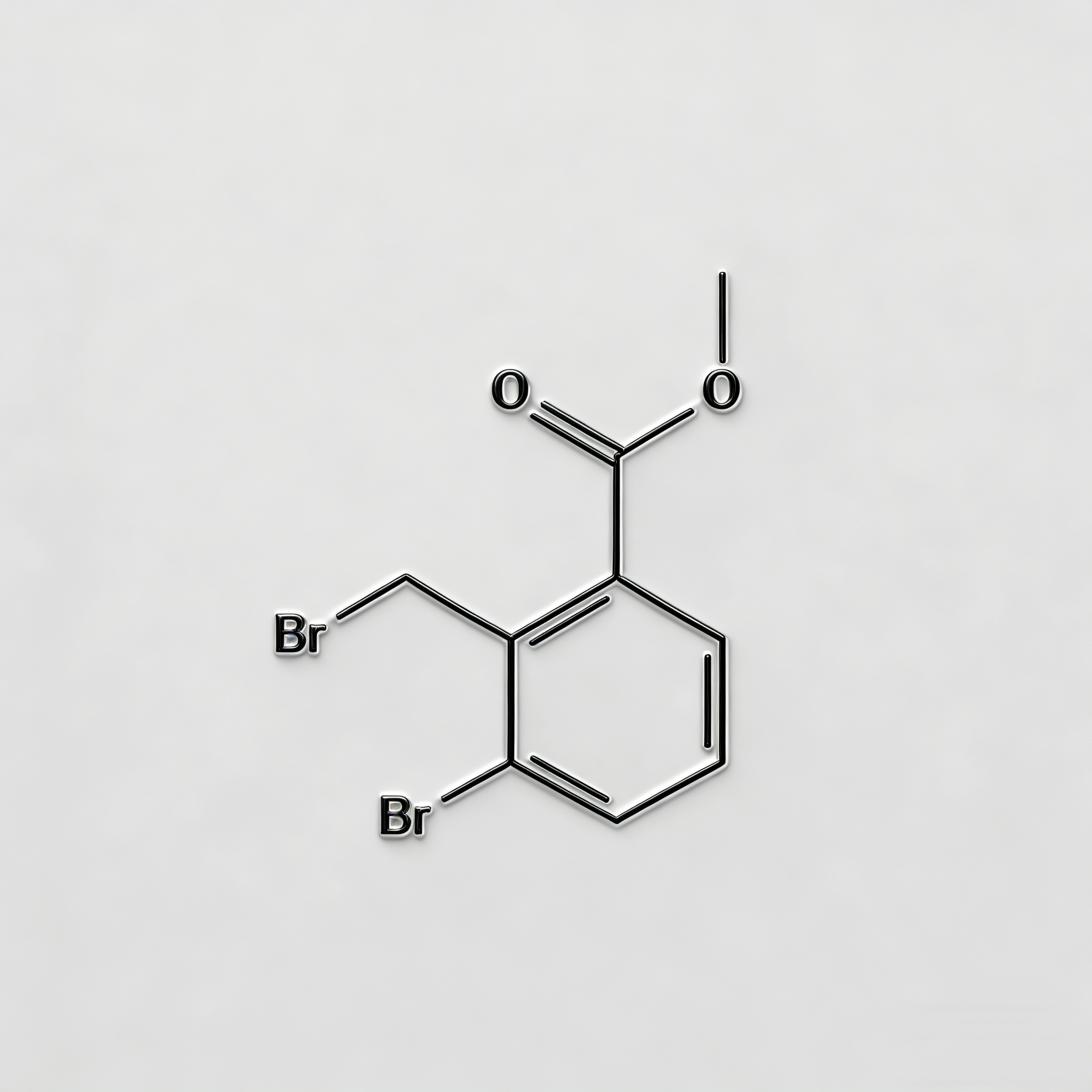
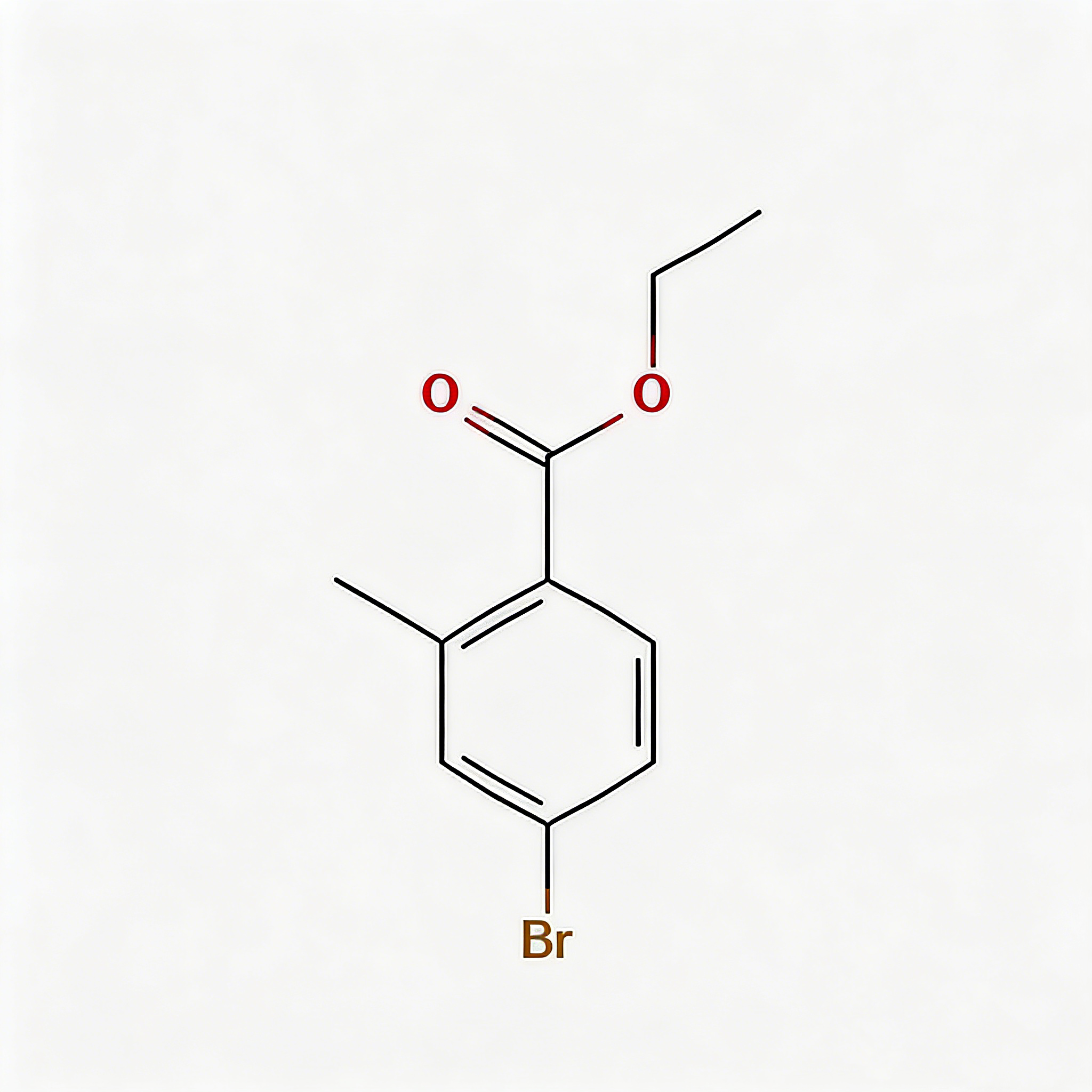
tert-Butyl 4-bromo-2-methylbenzoate
tert-Butyl 4-bromo-2-methylbenzoate (CAS 445003-37-2) is a strategically protected halogenated aromatic ester engineered for pharmaceutical intermediate synthesis, agrochemical development, and materials science applications. This molecule integrates a benzoate core—a benzene ring with a tert-butyl ester substituent—with a bromine atom at the C4 position and a methyl group at the C2 position. The tert-butyl ester moiety serves as an acid-labile protecting group for the carboxylic acid, enabling compatibility with base-sensitive reactions and orthogonal deprotection strategies (cleavage via TFA/HCl), while the C4-bromo substituent enables Pd-catalyzed cross-coupling (Suzuki, Stille, Negishi). The ortho-methyl group provides steric modulation and metabolic stability enhancement. Compared to its free acid analog (CAS 68837-59-2), this ester form offers improved lipophilicity and solubility in organic solvents. Compared to its methyl ester analog (CAS 78471-43-9 derivative), the tert-butyl group allows acid-mediated deprotection rather than base-mediated hydrolysis, providing synthetic flexibility. This intermediate is particularly valuable for constructing NSAID analogs, kinase inhibitor cores, and functional materials where controlled acid release and cross-coupling diversification are critical. Manufactured under controlled conditions, it supports drug discovery programs requiring modular access to differentially protected halogenated aromatic acids.
Property
|
Product Name
|
tert-Butyl 4-bromo-2-methylbenzoate
|
|
CAS Number
|
445003-37-2
|
|
Chinese Name
|
4-溴-2-甲基苯甲酸叔丁酯
|
|
Synonyms
|
tert-Butyl 4-bromo-2-methylbenzoate; 4-Bromo-2-methylbenzoic acid tert-butyl ester
|
|
Chinese Synonyms
|
4-溴-2-甲基苯甲酸叔丁酯; 4-溴邻甲苯甲酸叔丁酯
|
|
Molecular Formula
|
C₁₂H₁₅BrO₂
|
|
Molecular Weight
|
271.15 g/mol
|
|
Purity
|
≥ 98% (HPLC/GC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Benzoic Acid Derivatives / Acid-Labile Protected Esters
|
|
Appearance
|
Yellow to orange liquid
|
|
Melting Point
|
~60-65°C (estimated, literature reference recommended)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed. Protect from strong acids (TFA, HCl) to avoid premature ester deprotection and from strong bases to avoid hydrolysis.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
NSAID analog synthesis, kinase inhibitor development, Pd-catalyzed cross-coupling (C4-Br), acid-mediated deprotection studies, agrochemical intermediate synthesis, functional material precursor
|
|
Reactivity
|
Suitable for Pd-catalyzed cross-coupling (C4-Br displacement), ester deprotection (TFA/DCM or HCl/dioxane), electrophilic aromatic substitution at C3/C5/C6, and sequential functionalization
|
|
Special Note
|
✅ Acid-Labile Protected Ester—tert-butyl group enables orthogonal deprotection vs. methyl esters. C4-Br enables cross-coupling. Distinct from free acid (CAS 68837-59-2) by protection status. Part of our halogenated aromatic ester series.
|