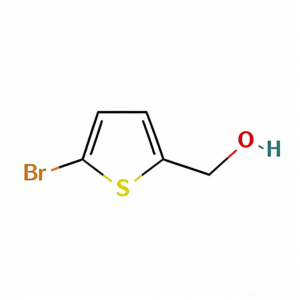
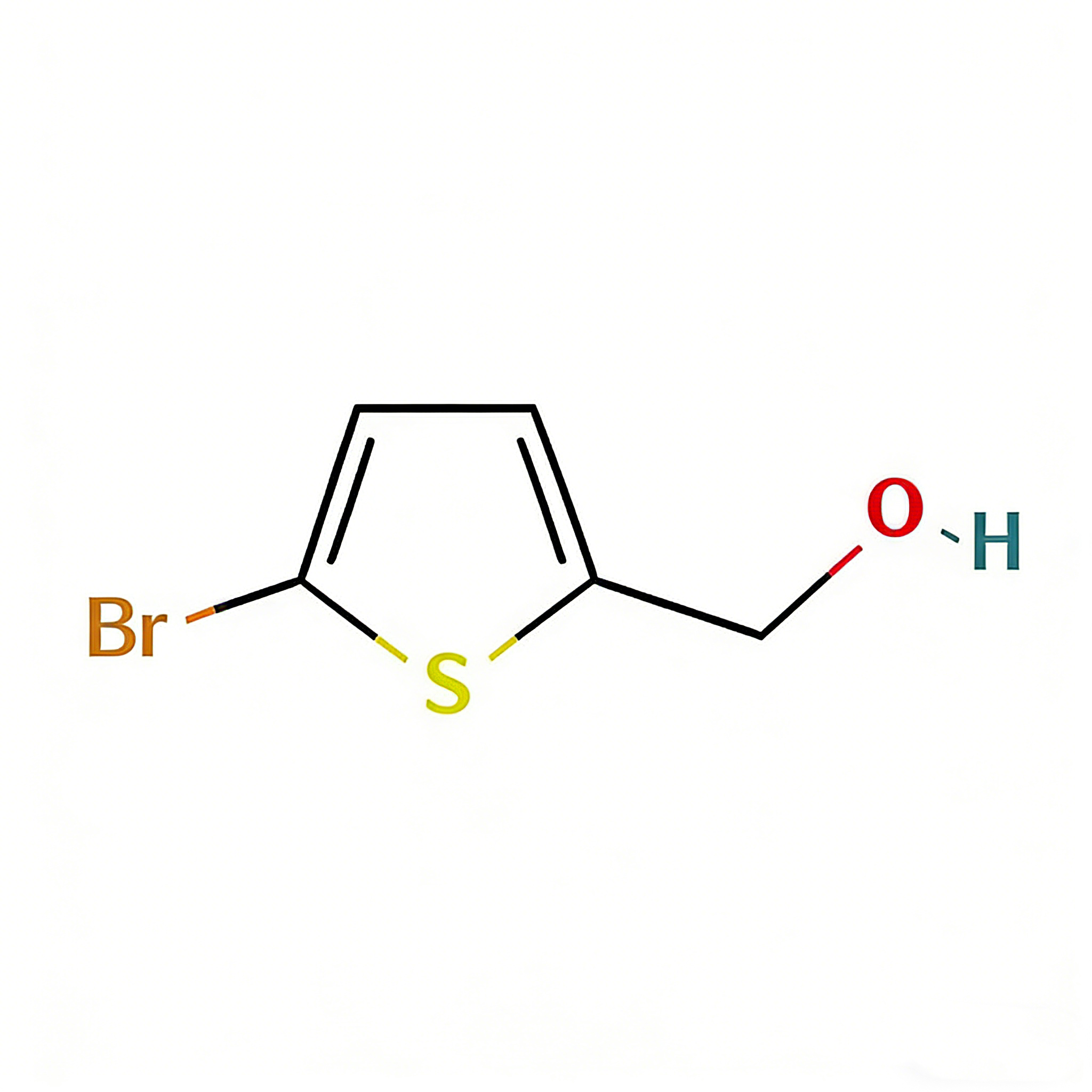
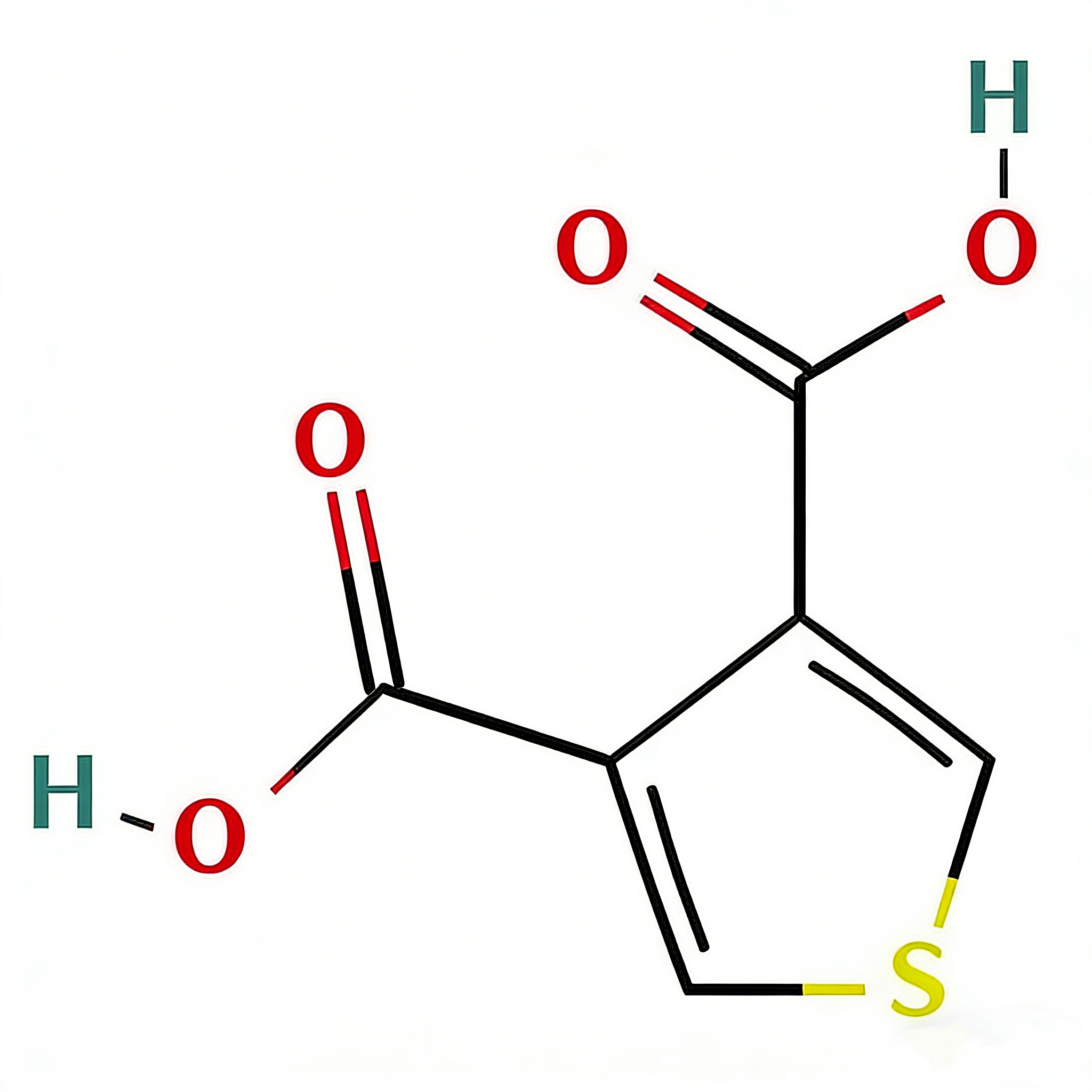
(5-Bromothien-2-yl)methanol
(5-Bromothien-2-yl)methanol (CAS 79387-71-6) is a strategically functionalized thiophene derivative engineered for cross-coupling reactions and heterocyclic diversification in medicinal chemistry and materials science. This molecule integrates a 2,5-disubstituted thiophene core—a linear conjugated system prevalent in conductive polymers and pharmaceuticals—with a bromine atom at the C5 position and a hydroxymethyl group at the C2 position. The C5-bromo handle serves as a privileged site for Pd-catalyzed cross-coupling (Suzuki, Stille, Negishi), enabling efficient extension of the π-conjugated system, while the C2-hydroxymethyl moiety provides a versatile handle for oxidation to aldehyde/acid, esterification, or conversion to halides for further functionalization. The 2,5-substitution pattern offers distinct electronic properties compared to 2,3- or 3-substituted analogs, facilitating linear charge transport in organic electronics and specific binding orientations in biological targets. This intermediate is particularly valuable for constructing thiophene-based oligomers, kinase inhibitor scaffolds, and agrochemical actives where the bromo-alcohol motif enables modular synthetic strategies. Manufactured under controlled conditions, it supports drug discovery and materials science programs requiring precise control over thiophene substitution patterns.
Property
|
Product Name
|
(5-Bromothien-2-yl)methanol
|
|
CAS Number
|
79387-71-6
|
|
Chinese Name
|
(5-溴噻吩 -2-基) 甲醇
|
|
Synonyms
|
5-Bromo-2-thienylmethanol; (5-Bromo-2-thienyl)methanol; C₅H₅BrOS heterocycle; 5-Bromothiophene-2-methanol
|
|
Chinese Synonyms
|
5-溴 -2-噻吩甲醇; 5-溴噻吩 -2-甲醇; 溴代噻吩甲醇; 2-羟甲基 -5-溴噻吩
|
|
Molecular Formula
|
C₅H₅BrOS
|
|
Molecular Weight
|
193.06 g/mol (calculated: C5×12.01 + H5×1.008 + Br×79.90 + O×16.00 + S×32.07)
|
|
Purity
|
≥ 98% (GC/HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Thiophene Derivatives / Brominated Alcohols
|
|
Appearance
|
Colorless to Pale Yellow Liquid
|
|
Boiling Point
|
~260-270°C at 760 mmHg (estimated)
|
|
Density
|
~1.75 g/cm³ (estimated)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong oxidizing agents to avoid alcohol oxidation or sulfur oxidation.
|
|
Shelf Life
|
18-24 months under recommended storage conditions
|
|
Packaging
|
100g/bottle, 500g/bottle, 1kg/bottle, 5kg/drum, or customized according to customer requirements
|
|
Applications
|
Thiophene oligomer synthesis (conducting polymers), kinase inhibitor scaffold development, cross-coupling reaction precursor (Suzuki/Stille), agrochemical active ingredient precursors, alcohol derivatization (oxidation/esterification)
|
|
Reactivity
|
Suitable for Pd-catalyzed cross-coupling (C5-Br), alcohol oxidation to aldehyde/acid, esterification, conversion to halides (for further coupling), and electrophilic aromatic substitution at C3/C4
|
|
Special Note
|
✅ Dual-Functionality Scaffold—C5-Br for coupling, C2-CH₂OH for derivatization. 2,5-substitution enables linear conjugation. Light-sensitive—brominated thiophenes may darken on exposure. Part of our thiophene building block series.
|