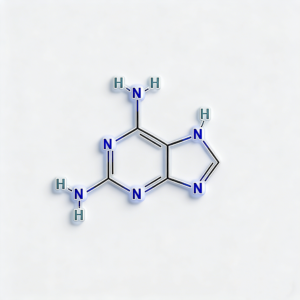
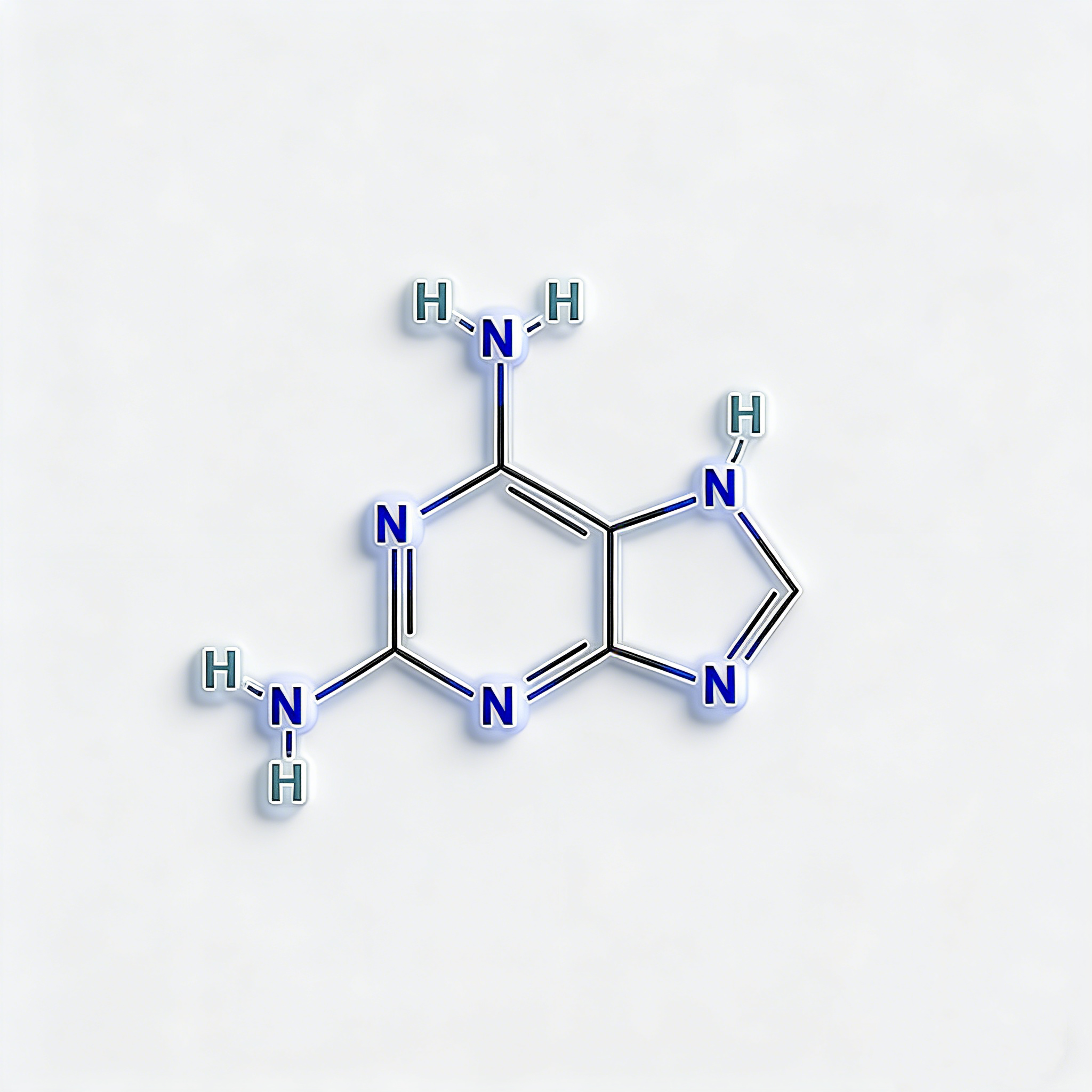
2,6-Diaminopurine
2,6-Diaminopurine (CAS 1904-98-9) is a high-purity nucleobase analog engineered for antiviral drug development, oligonucleotide research, and kinase inhibitor synthesis. This electron-rich purine scaffold features dual amino groups at C2 and C6 positions, enabling triple hydrogen-bonding capacity with thymine—offering enhanced binding affinity compared to natural adenine. The 2,6-diamino substitution pattern serves as a versatile platform for N9-glycosylation to access nucleoside analogs (e.g., ganciclovir precursors), while the amino groups enable selective acylation, alkylation, or diazotization for further diversification. This intermediate is particularly valuable for constructing antisense oligonucleotides with improved hybridization stability, antiviral therapeutics targeting viral DNA polymerases, and ATP-competitive kinase inhibitors where precise hydrogen-bonding geometry dictates target selectivity. Manufactured under controlled conditions, it supports drug discovery programs requiring modular access to functionalized purine architectures with enhanced base-pairing properties .
Property
|
Product Name
|
2,6-Diaminopurine
|
|
CAS Number
|
1904-98-9
|
|
Chinese Name
|
2,6-二氨基嘌呤
|
|
Synonyms
|
Purine, 2,6-diamino-; 2,6-Diamino-9H-purine; 2-Aminoadenine; C₅H₆N₆ heterocycle; DAP
|
|
Chinese Synonyms
|
2,6-二氨基 -9H-嘌呤; 2-氨基腺嘌呤; 二氨基嘌呤; 嘌呤 -2,6-二胺
|
|
Molecular Formula
|
C₅H₆N₆
|
|
Molecular Weight
|
150.14 g/mol
|
|
Purity
|
≥ 98% (HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Purine Derivatives / Nucleobase Analogs
|
|
Appearance
|
White to Off-white Crystalline Powder
|
|
Melting Point
|
>360°C (decomposition, literature reference recommended)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed. Protect from strong oxidizing agents to avoid amino group oxidation.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Antiviral nucleoside analog synthesis, antisense oligonucleotide stabilization, kinase inhibitor scaffold development, DNA/RNA base-pairing research, heterocyclic fusion reactions, acylation/alkylation at amino positions
|
|
Reactivity
|
Suitable for N9-glycosylation, amino group acylation/alkylation, diazotization, heterocyclic ring fusion, and electrophilic aromatic substitution
|
|
Special Note
|
Triple H-Bond Base Analog—forms 3 H-bonds with Thymine (vs. Adenine’s 2). Electron-rich scaffold contrasts with electrophilic 2,6-dichloropurine (CAS 5451-40-1). Ideal for high-affinity binding applications. Part of our purine building block series.
|