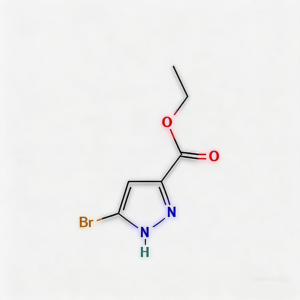
Ethyl 3-bromo-1h-pyrazole-5-carboxylate
Ethyl 3-bromo-1H-pyrazole-5-carboxylate (CAS 1886994-07-5) is a high-purity, regiochemically defined heterocyclic building block widely used in pharmaceutical intermediates and medicinal chemistry . This strategically brominated pyrazole ester features two orthogonal reactive handles: a C3-bromine atom for diverse palladium-catalyzed cross-coupling reactions, and a C5-ethyl ester for hydrolysis or amide coupling . The unique 3,5-substitution pattern creates a distinct electronic distribution across the pyrazole ring, making this regioisomeric scaffold particularly valuable for constructing kinase inhibitors, ERK inhibitors for regulating cell proliferation and apoptosis, and agrochemical active ingredients where precise positional control dictates binding orientation in target pockets . Manufactured under controlled conditions with purity ≥95-98% , this intermediate supports drug discovery programs requiring modular diversification of nitrogen-rich heterocyclic architectures with precise regiochemical control.
Property
|
Product Name
|
Ethyl 3-bromo-1H-pyrazole-5-carboxylate
|
|
CAS Number
|
1886994-07-5
|
|
Chinese Name
|
3-溴-1H-吡唑 -5-羧酸乙酯
|
|
Synonyms
|
Ethyl 3-bromopyrazole-5-carboxylate; 1H-Pyrazole-5-carboxylic acid, 3-bromo-, ethyl ester; 3-Bromo-5-ethoxycarbonylpyrazole; C₆H₇BrN₂O₂ heterocycle; ethyl 5-bromo-1H-pyrazole-3-carboxylate
|
|
Chinese Synonyms
|
3-溴吡唑 -5-羧酸乙酯; 3-溴-5-乙氧羰基吡唑; 溴代吡唑羧酸乙酯; 3-溴-1H-吡唑 -5-甲酸乙酯
|
|
Molecular Formula
|
C₆H₇BrN₂O₂
|
|
Molecular Weight
|
219.04 g/mol
|
|
Purity
|
≥ 98% (GC/HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Pyrazole Derivatives / Brominated Heterocyclic Esters
|
|
Appearance
|
White to pale yellow solid
|
|
Melting Point
|
~150-155°C (estimated, literature reference recommended)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong bases to avoid ester hydrolysis or debromination side reactions.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Kinase inhibitor synthesis via C3 cross-coupling, fused heterocycle construction (regioselective cyclization), agrochemical active ingredient precursors, SAR exploration via bromine displacement, prodrug design via ester modification
|
|
Reactivity
|
Suitable for Suzuki coupling, Buchwald-Hartwig amination, Sonogashira coupling (C3-Br displacement), ester hydrolysis to acid, amide coupling, electrophilic aromatic substitution at C4, and pyrazole N1-derivatization
|
|
Special Note
|
✅ 3-Bromo-5-Carboxylate Regioisomer—distinct electronic profile vs. 5-bromo-3-carboxylate analogs (CAS 1328893-17-9). The C3-Br position offers unique coupling kinetics. Ethyl ester provides enhanced lipophilicity vs. methyl analogs. Part of our brominated heterocyclic building block series.
|