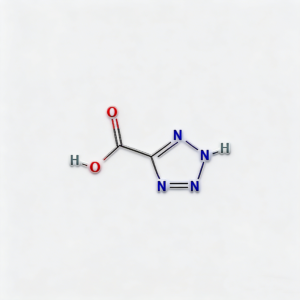
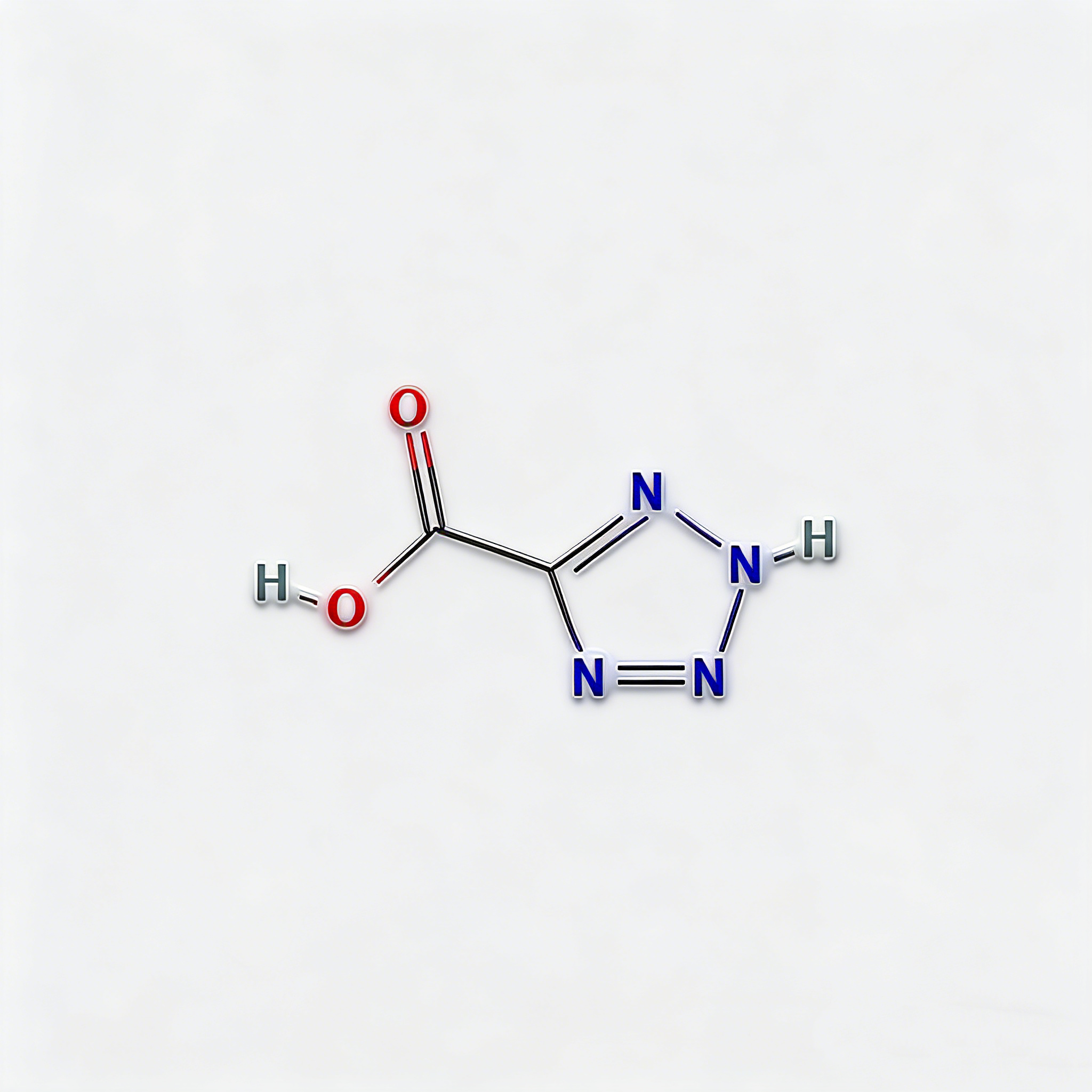
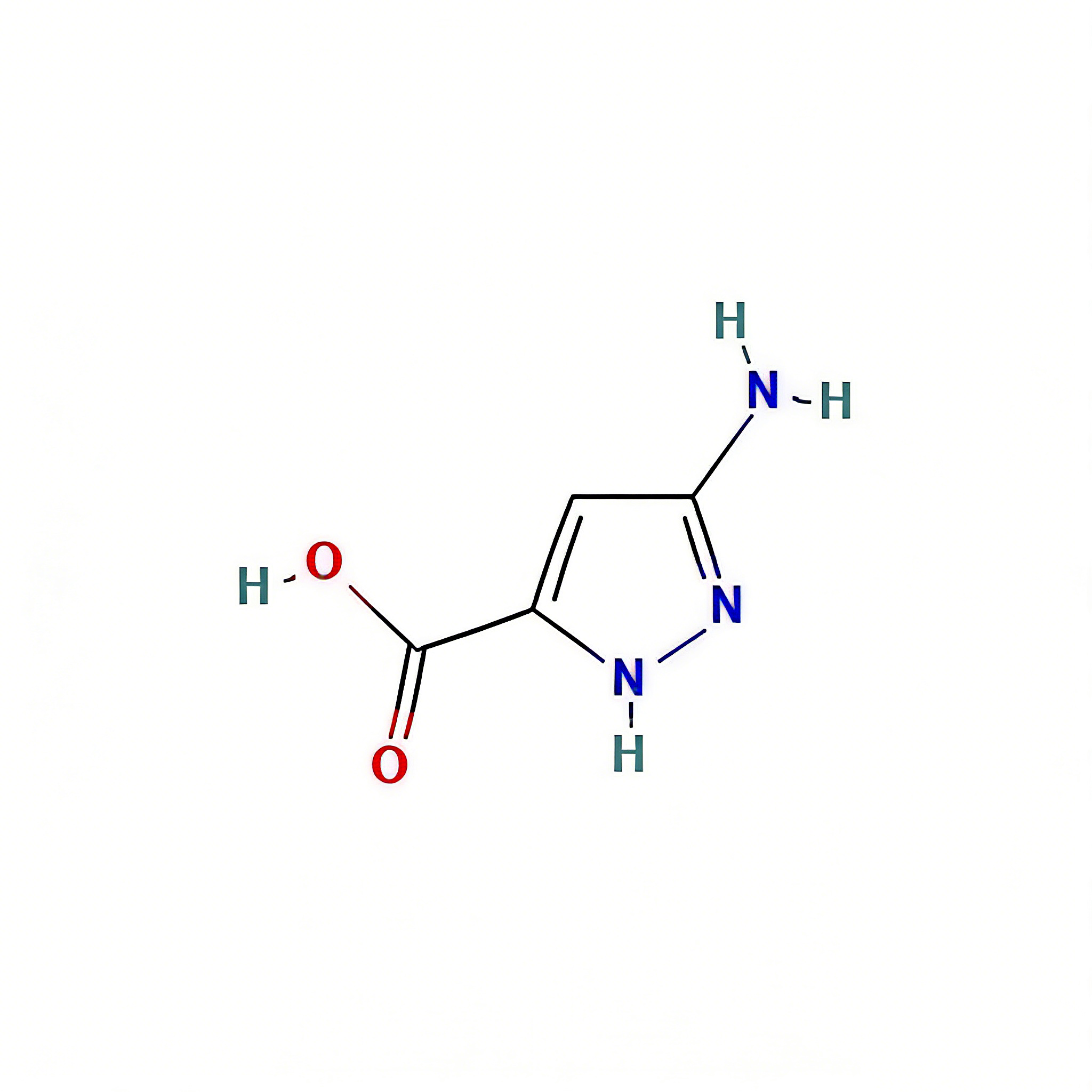
2H-1,2,3,4-tetrazole-5-carboxylic acid
2H-1,2,3,4-Tetrazole-5-carboxylic acid (CAS 75773-99-8) is a high-purity, nitrogen-rich heterocyclic building block engineered for bioisosteric applications in medicinal chemistry and agrochemical development . This molecule integrates a 1,2,3,4-tetrazole core—a pentazole ring system with exceptional metabolic stability and hydrogen-bonding capacity—with a carboxylic acid substituent at the C5 position. The tetrazole ring serves as a privileged bioisostere for carboxylic acids, cis-amides, and oxadiazoles, offering enhanced metabolic stability, improved membrane permeability, and unique electronic properties compared to conventional aromatic systems . The C5-carboxylic acid moiety enables direct amide coupling, salt formation for solubility enhancement, or decarboxylative cross-coupling strategies. This intermediate is particularly valuable for constructing kinase inhibitor cores, angiotensin II receptor antagonists for hypertension treatment, and agrochemical actives where the tetrazole-carboxylate motif optimizes target engagement and pharmacokinetic profiles . Manufactured under controlled conditions , it supports drug discovery programs requiring modular access to nitrogen-dense heterocyclic architectures with tunable acidity and hydrogen-bonding geometry.
Property
|
Product Name
|
2H-1,2,3,4-Tetrazole-5-carboxylic acid
|
|
CAS Number
|
75773-99-8
|
|
Chinese Name
|
2H-1,2,3,4-四唑 -5-羧酸
|
|
Synonyms
|
1,2,3,4-Tetrazole-5-carboxylic acid; 5-Carboxy-2H-tetrazole; C₂H₂N₄O₂ heterocycle; Tetrazole-5-carboxylate
|
|
Chinese Synonyms
|
1,2,3,4-四唑 -5-羧酸; 5-羧基 -2H-四唑; 四唑羧酸; 2H-四唑 -5-甲酸
|
|
Molecular Formula
|
C₂H₂N₄O₂
|
|
Molecular Weight
|
114.06 g/mol
|
|
Purity
|
≥ 98% (HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates; Tetrazole Derivatives; Bioisosteric Building Blocks; Heterocyclic Carboxylic Acids
|
|
Appearance
|
White to off-white solid
|
|
Melting Point
|
~180-190°C (decomposition, literature reference recommended)
|
|
pKa
|
~4.5-5.0 (estimated, tetrazole NH + carboxylic acid)
|
|
Storage Conditions
|
Store at 2-8°C under inert atmosphere in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed. Protect from strong oxidizing agents and bases to avoid ring degradation or decarboxylation.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Bioisosteric replacement for carboxylic acids in drug design, kinase inhibitor synthesis, angiotensin receptor antagonist development, agrochemical active ingredient precursors, salt formation for solubility enhancement, decarboxylative cross-coupling strategies
|
|
Reactivity
|
Suitable for amide coupling (activation via EDC/HATU), esterification, salt formation with organic/inorganic bases, decarboxylative coupling, tetrazole N-alkylation, and bioisosteric scaffold hopping
|
|
Special Note
|
Nitrogen-Rich Bioisostere—tetrazole core offers superior metabolic stability vs. conventional aromatics. Dual acidic protons (tetrazole NH + COOH) enable versatile salt formation. Part of our high-nitrogen heterocyclic series.
|