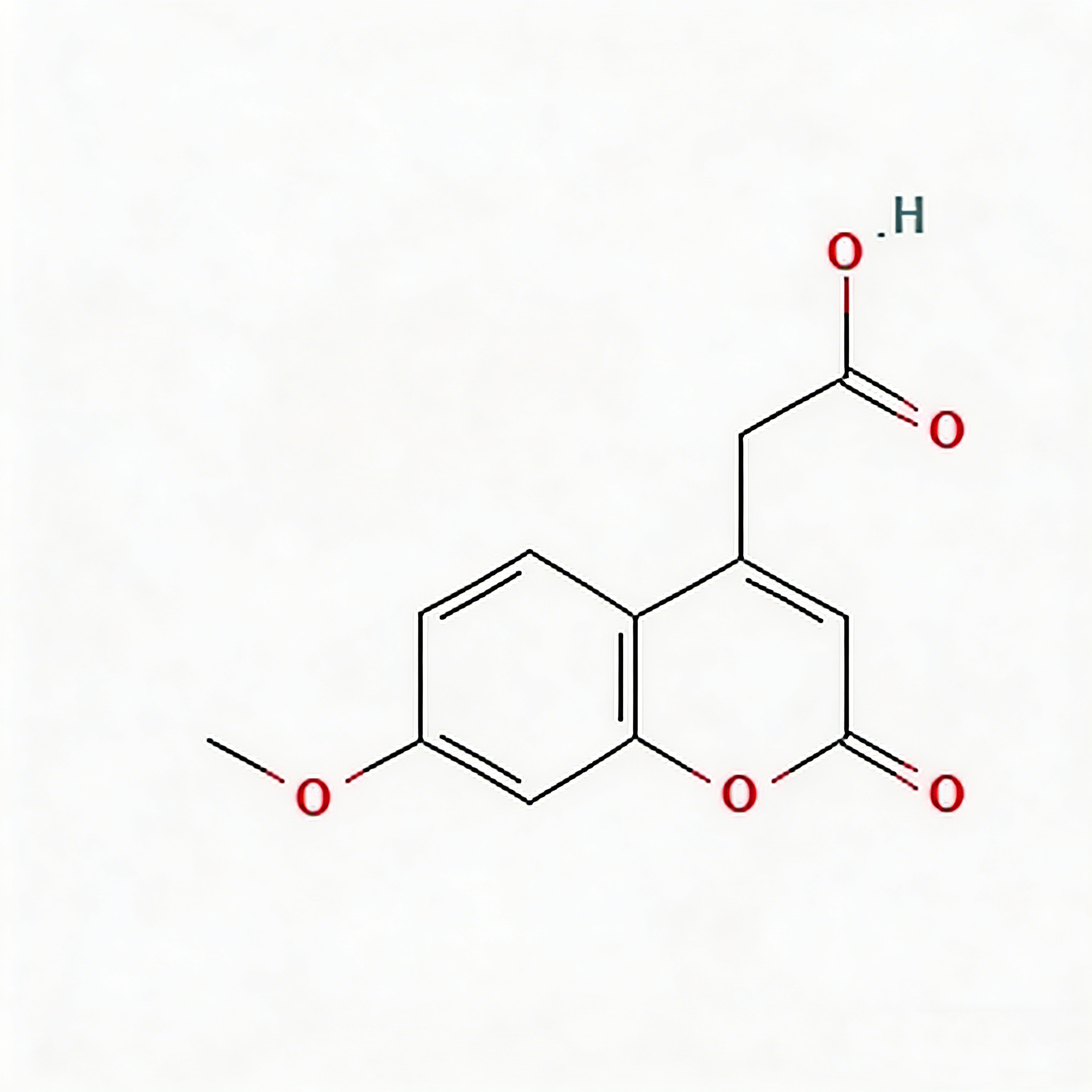
3,4-Dihydro-1h-2-benzopyran-3-carboxylic acid
3,4-Dihydro-1H-2-benzopyran-3-carboxylic acid (CAS 1261578-13-5) is a strategically functionalized isochroman scaffold engineered for chiral drug discovery and heterocyclic diversification in medicinal chemistry. This molecule integrates a 3,4-dihydro-1H-2-benzopyran (isochroman) core—a benzene-fused oxygen heterocycle with conformational rigidity—with a carboxylic acid substituent at the C3 position, which serves as a chiral center enabling enantioselective synthesis. The fused bicyclic architecture provides a planar aromatic domain for π-stacking interactions alongside a flexible saturated pyran ring for conformational tuning, while the C3-carboxylic acid moiety enables direct amide coupling, salt formation, or decarboxylative functionalization strategies. This intermediate is particularly valuable for constructing kinase inhibitor cores, CNS-penetrant scaffolds, and GPCR modulators where the isochroman-carboxylate motif optimizes target engagement through defined stereochemistry and hydrogen-bonding geometry. Manufactured under controlled conditions, it supports drug discovery programs requiring modular access to chiral oxygen heterocycles with tunable lipophilicity and metabolic stability.
Property
|
Product Name
|
3,4-Dihydro-1H-2-benzopyran-3-carboxylic acid
|
|
CAS Number
|
1261578-13-5
|
|
Chinese Name
|
3,4-二氢 -1H-2-苯并吡喃 -3-羧酸
|
|
Synonyms
|
Isochroman-3-carboxylic acid; 3-Carboxyisochroman; 3,4-Dihydroisochromene-3-carboxylic acid; C₁₀H₁₀O₃ heterocycle
|
|
Chinese Synonyms
|
异色满 -3-羧酸; 3-羧基异色满; 3,4-二氢异色烯 -3-羧酸; 二氢苯并吡喃羧酸
|
|
Molecular Formula
|
C₁₀H₁₀O₃
|
|
Molecular Weight
|
178.18 g/mol
|
|
Purity
|
≥ 98% (HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates; Chromane Derivatives; Chiral Carboxylic Acids; Oxygen Heterocycles
|
|
Appearance
|
White to off-white solid
|
|
Melting Point
|
~165-175°C (estimated, literature reference recommended; may exist as racemate)
|
|
Storage Conditions
|
Store at 2-8°C under inert atmosphere in a cool, dry, well-ventilated area away from light and moisture.Keep container tightly closed. Protect from strong oxidizing agents and bases to avoid decarboxylation or ring oxidation.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Chiral kinase inhibitor synthesis, CNS drug scaffold development, GPCR modulator design, amide coupling for peptide mimetics, salt formation for solubility enhancement, decarboxylative cross-coupling strategies
|
|
Reactivity
|
Suitable for amide coupling (activation via EDC/HATU), esterification, salt formation, decarboxylative coupling, electrophilic aromatic substitution on benzene ring, and chiral resolution at C3 stereocenter
|
|
Special Note
|
Chiral Isochroman Scaffold—C3 carboxylic acid creates a stereogenic center enabling enantioselective drug design. Fused benzene+pyran architecture balances rigidity and flexibility. Part of our chiral oxygen heterocycle series.
|