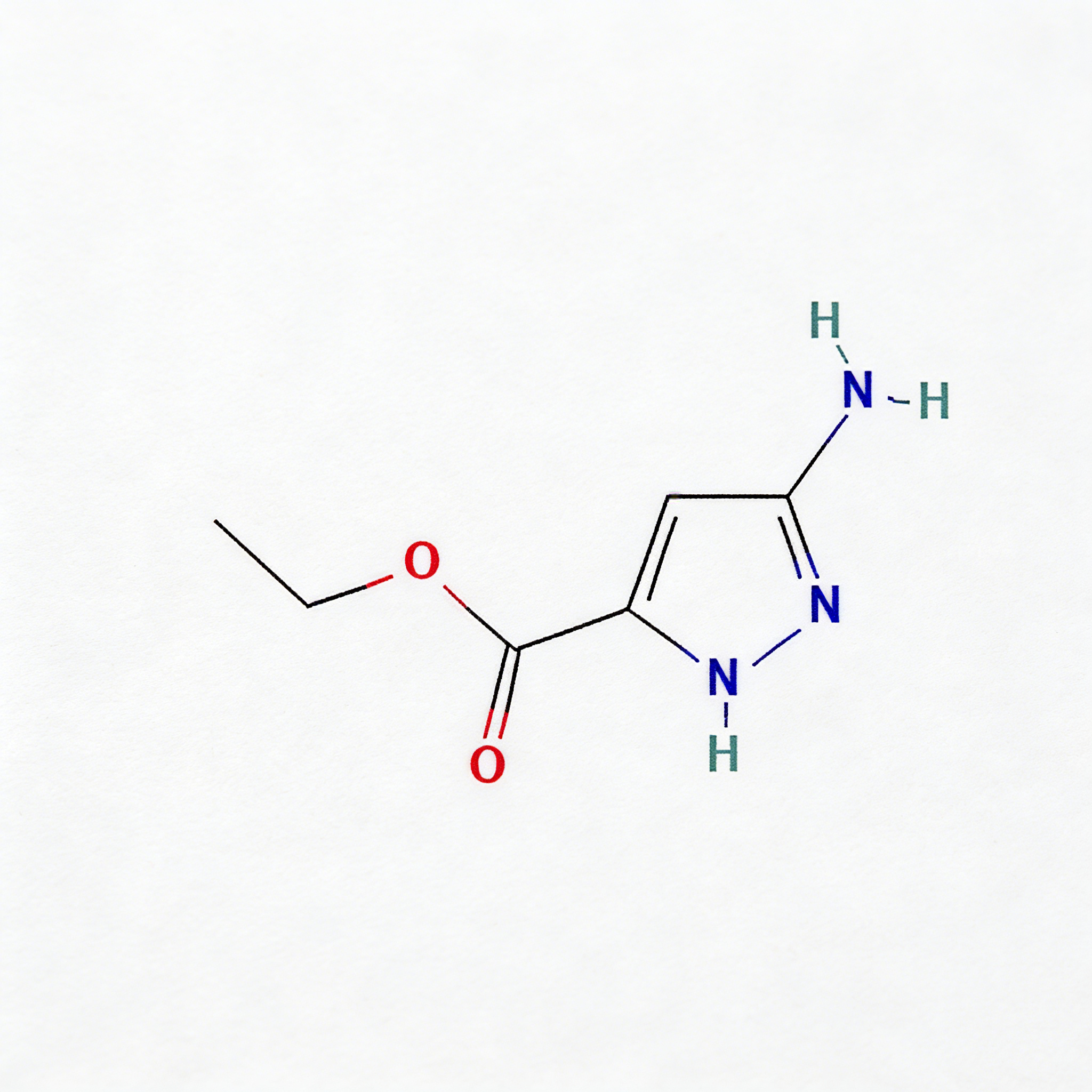
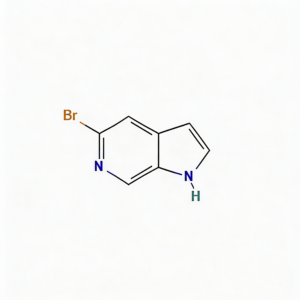
5-Bromo-1h-pyrrolo[2,3-c]pyridine
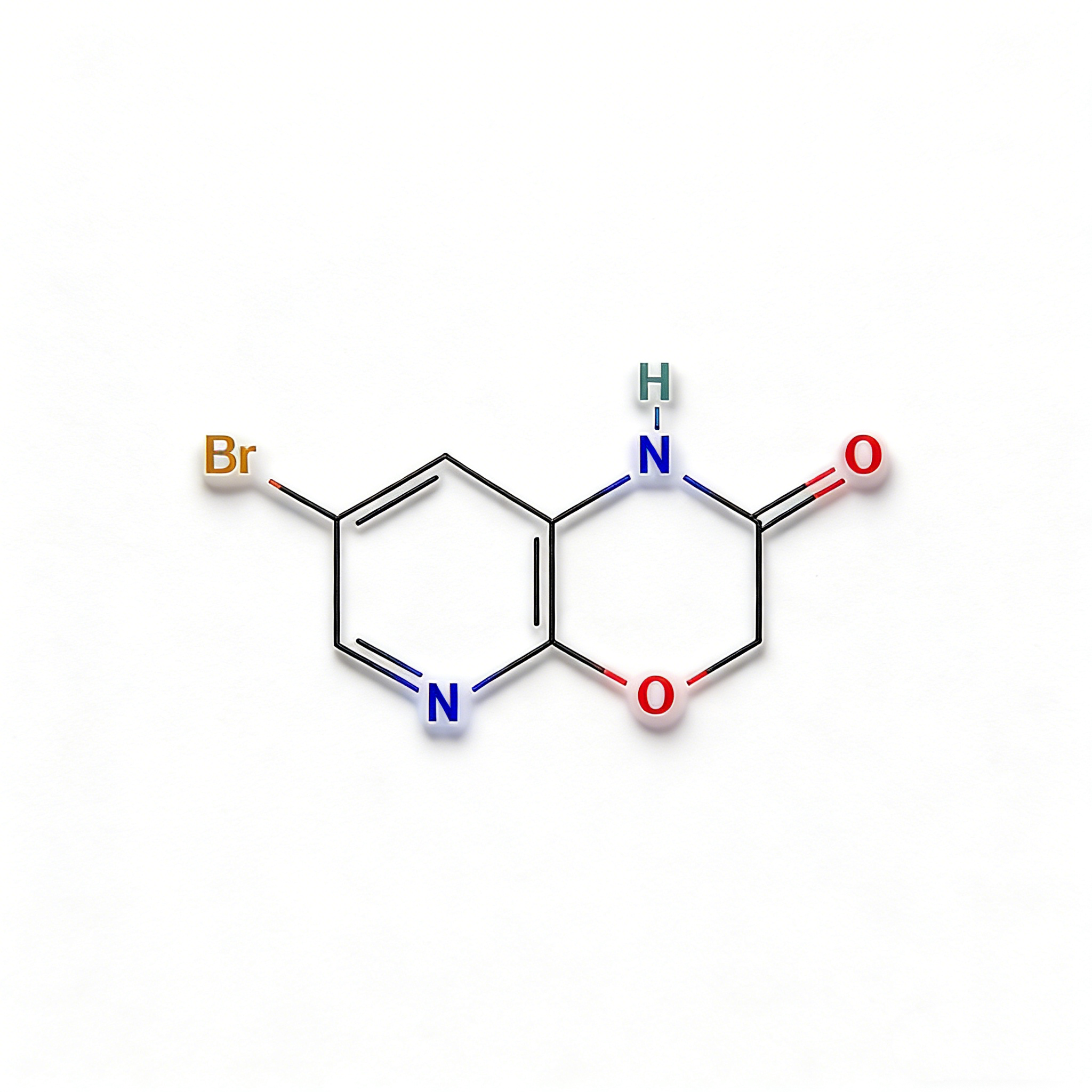
5-Bromo-1H-pyrrolo[2,3-c]pyridine (CAS 1215387-58-8) is a strategically brominated fused bicyclic scaffold engineered for kinase inhibitor development and heterocyclic diversification in medicinal chemistry. This molecule integrates a pyrrolo[2,3-c]pyridine core—a nitrogen-rich 7-azaindole isostere—with a bromine substituent at the C5 position. The fused 5,6-bicyclic system provides a planar, rigid architecture ideal for π-stacking interactions in ATP-binding pockets, while the C5-bromo handle enables efficient Pd-catalyzed cross-coupling (Suzuki, Buchwald-Hartwig, Sonogashira) to introduce diverse aryl, heteroaryl, or amine substituents. Distinct from the more common pyrrolo[2,3-b]pyridine (7-azaindole) isomers, the [2,3-c] fusion pattern alters the electronic distribution and hydrogen-bonding geometry, offering unique selectivity profiles against kinase targets. This intermediate is particularly valuable for constructing targeted oncology therapeutics, CNS-penetrant scaffolds, and agrochemical actives where fused nitrogen heterocycles enhance metabolic stability and binding affinity. Manufactured with purity ≥98% under controlled conditions, it supports drug discovery programs requiring modular access to azaindole-like architectures with precise nitrogen positioning.
Property
|
Product Name
|
5-Bromo-1H-pyrrolo[2,3-c]pyridine
|
|
CAS Number
|
1215387-58-8
|
|
Chinese Name
|
5-溴 -1H-吡咯并 [2,3-c] 吡啶
|
|
Synonyms
|
5-Bromo-1H-pyrrolo[2,3-c]pyridine; 1H-Pyrrolo[2,3-c]pyridine, 5-bromo-; C₇H₅BrN₂ fused heterocycle
|
|
Chinese Synonyms
|
5-溴吡咯并吡啶; 5-溴 -1H-吡咯并 [2,3-c] 吡啶; 溴代氮杂吲哚类似物
|
|
Molecular Formula
|
C₇H₅BrN₂
|
|
Molecular Weight
|
197.03 g/mol
|
|
Purity
|
≥ 98% (HPLC/GC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Fused Heterocycles / Brominated Azaindoles
|
|
Appearance
|
Light brown to brown solid
|
|
Melting Point
|
~240-250°C (estimated, decomposition possible, literature reference recommended)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong bases to avoid debromination or ring degradation.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
customized according to customer requirements
|
|
Applications
|
Kinase inhibitor synthesis (ATP-mimetic scaffolds), oncology drug development, CNS-penetrant scaffold design, agrochemical active ingredient precursors, heterocyclic library synthesis via C5 coupling
|
|
Reactivity
|
Suitable for Suzuki coupling, Buchwald-Hartwig amination, Sonogashira coupling (C5-Br displacement), N1-alkylation/acylation, electrophilic aromatic substitution at C3/C4, and metal-catalyzed C–H functionalization
|
|
Special Note
|
Fused Bicyclic Scaffold—distinct electronic profile vs. pyrrolo[2,3-b]pyridine (7-azaindole) isomers. The C5-Br position offers strategic diversification. Part of our fused nitrogen heterocycle series.
|

![5-Bromo-1h-pyrrolo[2,3-c]pyridine 1215387-58-8](https://wealsunbio.com/wp-content/uploads/2026/01/1215387-58-8.png)