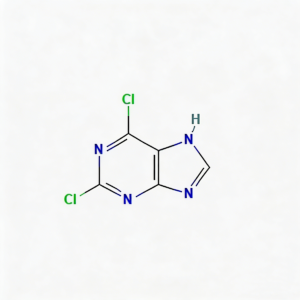
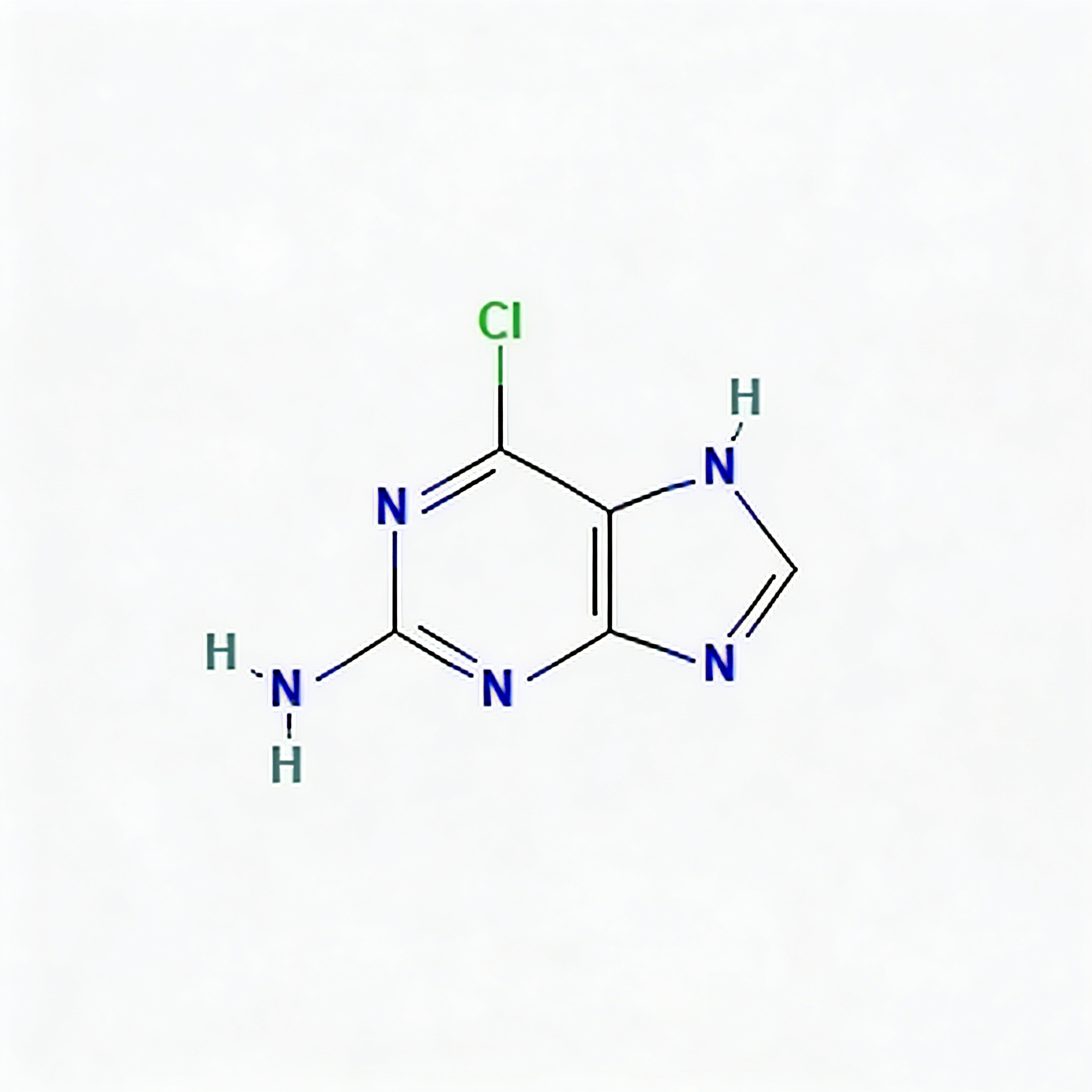
2,6-Dichloropurine
2,6-Dichloropurine (CAS 5451-40-1) is a highly reactive dual-halogenated purine scaffold engineered for sequential functionalization in nucleoside analog synthesis and kinase inhibitor development. Distinct from mono-chlorinated or amino-substituted purines, this molecule features chlorine substituents at both the C2 and C6 positions of the purine core, enabling two-stage diversification strategies. The C6-chlorine typically exhibits higher reactivity towards nucleophilic displacement, allowing selective mono-substitution followed by secondary modification at C2 under optimized conditions. This orthogonal reactivity profile makes it a privileged building block for constructing complex purine libraries, antiviral prodrugs, and ATP-competitive kinase inhibitors where precise control over substitution patterns is critical. The electron-deficient purine ring, enhanced by dual electron-withdrawing chloro groups, facilitates efficient coupling while maintaining metabolic stability in the final API. Manufactured with purity ≥98% under controlled conditions, it supports medicinal chemistry programs requiring modular access to multi-substituted purine architectures.
Property
|
Product Name
|
2,6-Dichloropurine
|
|
CAS Number
|
5451-40-1
|
|
Chinese Name
|
2,6-二氯嘌呤
|
|
Synonyms
|
Purine, 2,6-dichloro-; 2,6-Dichloro-9H-purine; C₅H₂Cl₂N₄ heterocycle; DCP
|
|
Chinese Synonyms
|
2,6-二氯 -9H-嘌呤; 二氯嘌呤; 2,6-二氯嘌呤碱; 嘌呤-2,6-二氯
|
|
Molecular Formula
|
C₅H₂Cl₂N₄
|
|
Molecular Weight
|
189.01 g/mol (calculated: C5×12.01 + H2×1.008 + Cl2×35.45 + N4×14.01)
|
|
Purity
|
≥ 98% (HPLC/GC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Purine Derivatives / Dihalo heterocycles
|
|
Appearance
|
White to Off-white Crystalline Powder
|
|
Melting Point
|
178-180°C
|
|
Storage Conditions
|
Store at 2-8°C under inert atmosphere, protected from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong bases and nucleophiles to avoid premature substitution or hydrolysis.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Sequential nucleophilic substitution, nucleoside analog synthesis (antiviral/anticancer), kinase inhibitor library construction, purine receptor modulator research, heterocyclic scaffold diversification
|
|
Reactivity
|
Suitable for stepwise nucleophilic aromatic substitution (C6 then C2), N9-glycosylation, cross-coupling reactions, and selective amination/alkoxylation/thiolation
|
|
Special Note
|
Dual-Reactivity Scaffold—enables sequential substitution at C2 and C6. More electrophilic than 2-amino-6-chloropurine (CAS 10310-21-1). Ideal for constructing asymmetrically substituted purines. Part of our purine building block series.
|