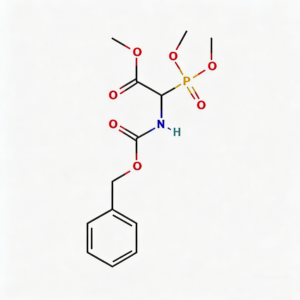
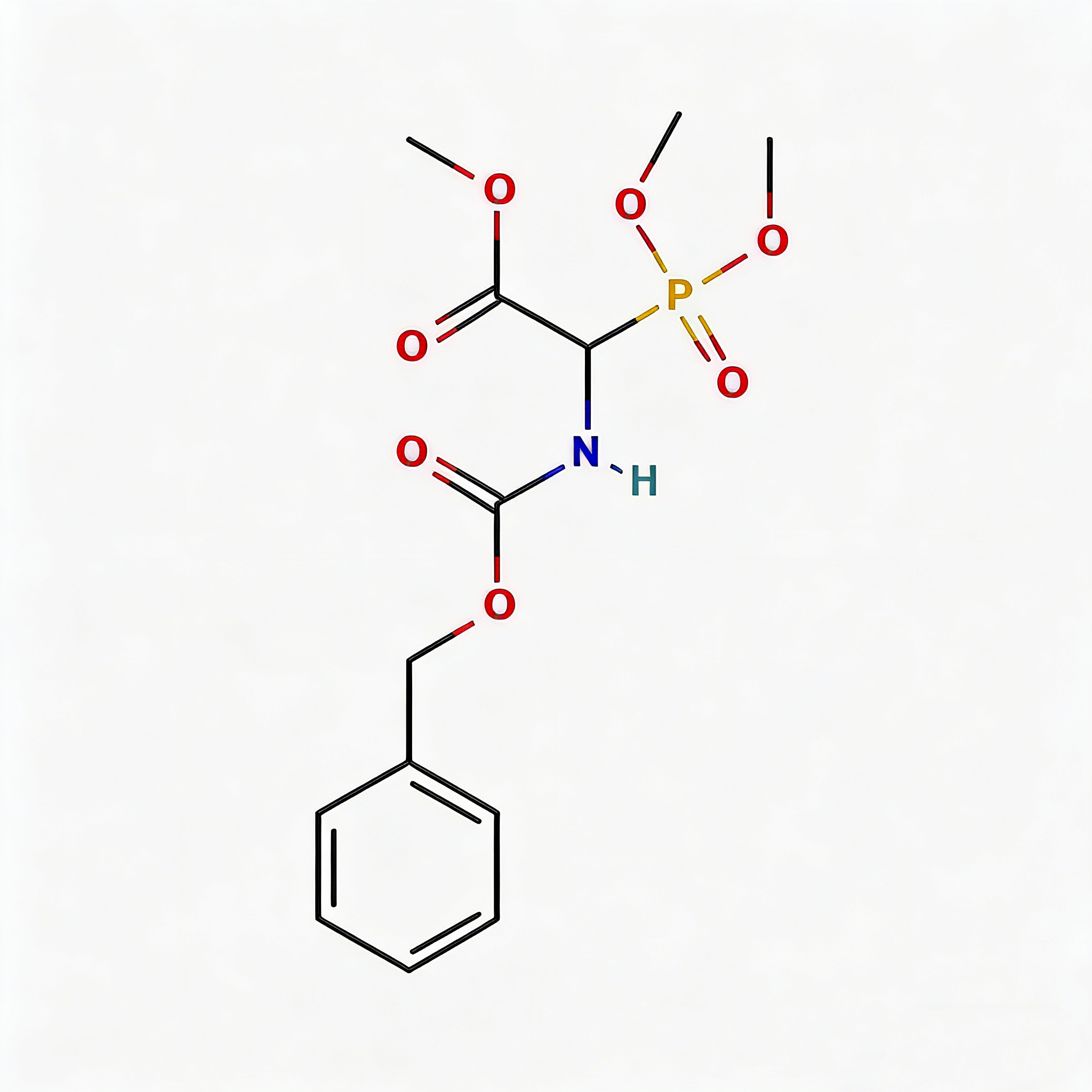
Benzyloxycarbonyl-alpha-phosphonoglycine trimethyl ester
Benzyloxycarbonyl-α-phosphonoglycine trimethyl ester (CAS 88568-95-0) is a strategically engineered phosphonate amino acid derivative designed for peptide mimetic development and transition-state analog research. This molecule integrates three orthogonal functional modules: a Cbz (benzyloxycarbonyl) protecting group enabling selective amine deprotection via hydrogenolysis, a phosphonate moiety at the α-carbon that mimics phosphate transition states with enhanced metabolic stability, and a trimethyl ester configuration that balances lipophilicity for improved membrane permeability. The phosphonate group—structurally analogous to phosphate but resistant to phosphatase cleavage—positions this intermediate as a privileged scaffold for protease inhibitor design, prodrug systems, and phosphonopeptide therapeutics targeting metalloenzymes. Manufactured under controlled conditions, it supports medicinal chemistry programs where phosphate mimicry is critical for binding affinity in kinase pathways, viral proteases, and bone metabolism modulators.
Property
|
Product Name
|
Benzyloxycarbonyl-α-phosphonoglycine trimethyl ester
|
|
CAS Number
|
88568-95-0
|
|
Chinese Name
|
苄氧羰基-α-膦酰基甘氨酸三甲酯
|
|
Synonyms
|
Cbz-α-phosphonoglycine trimethyl ester; Z-α-phosphonoglycine trimethyl ester; MFCD02685914
|
|
Chinese Synonyms
|
Cbz-α-膦酰甘氨酸三甲酯; Z-α-膦酰基甘氨酸三甲酯; 苄氧羰基氨基膦酰乙酸甲酯; 膦酰氨基酸衍生物
|
|
Molecular Formula
|
C₁₃H₁₈NO₇P
|
|
Molecular Weight
|
331.26 g/mol
|
|
Purity
|
≥ 98% (HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Phosphonate Amino Acids / Protected Building Blocks
|
|
Appearance
|
White to Off-white Crystalline Powder
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen or argon). Protect from strong acids/bases to avoid ester hydrolysis or premature Cbz deprotection.
|
|
Shelf Life
|
18-24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Phosphonopeptide synthesis, protease inhibitor development, prodrug design, transition-state analog research, metalloenzyme targeting, kinase pathway modulation, antiviral drug intermediates, bone resorption inhibitor precursors
|
|
Reactivity
|
Suitable for selective Cbz deprotection (catalytic hydrogenolysis), phosphonate ester hydrolysis to mono/di-acid forms, amide coupling at the carboxylate, phosphonate-directed metal coordination, and orthogonal deprotection strategies
|
|
Special Note
|
✅ Moisture-sensitive phosphonate ester—handle under inert atmosphere. Orthogonal deprotection: Cbz removable via H₂/Pd-C, methyl esters via controlled hydrolysis. Part of our phosphonate building block series.
|