3-Bromo-1h-1,2,4-triazole
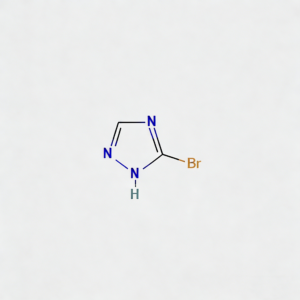
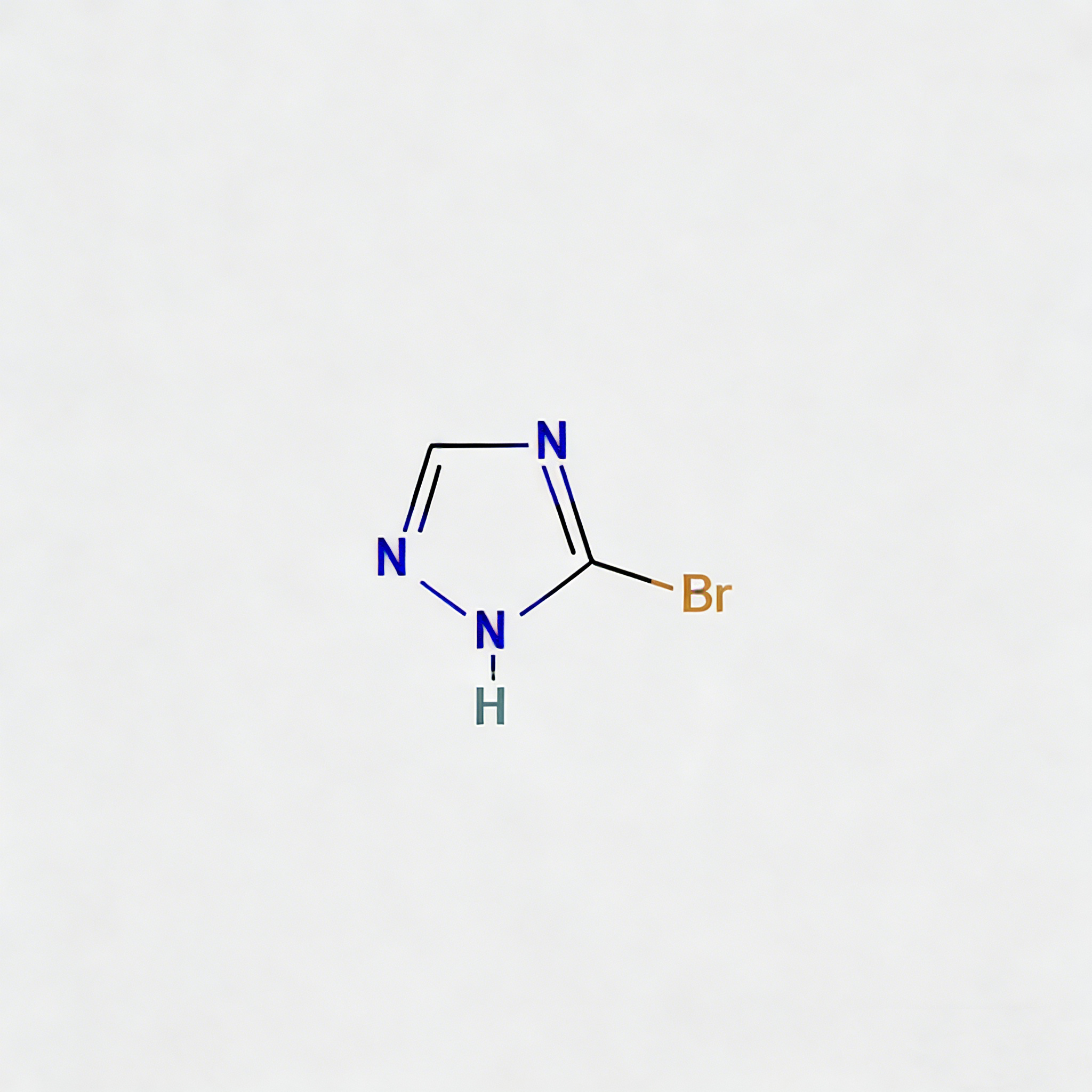
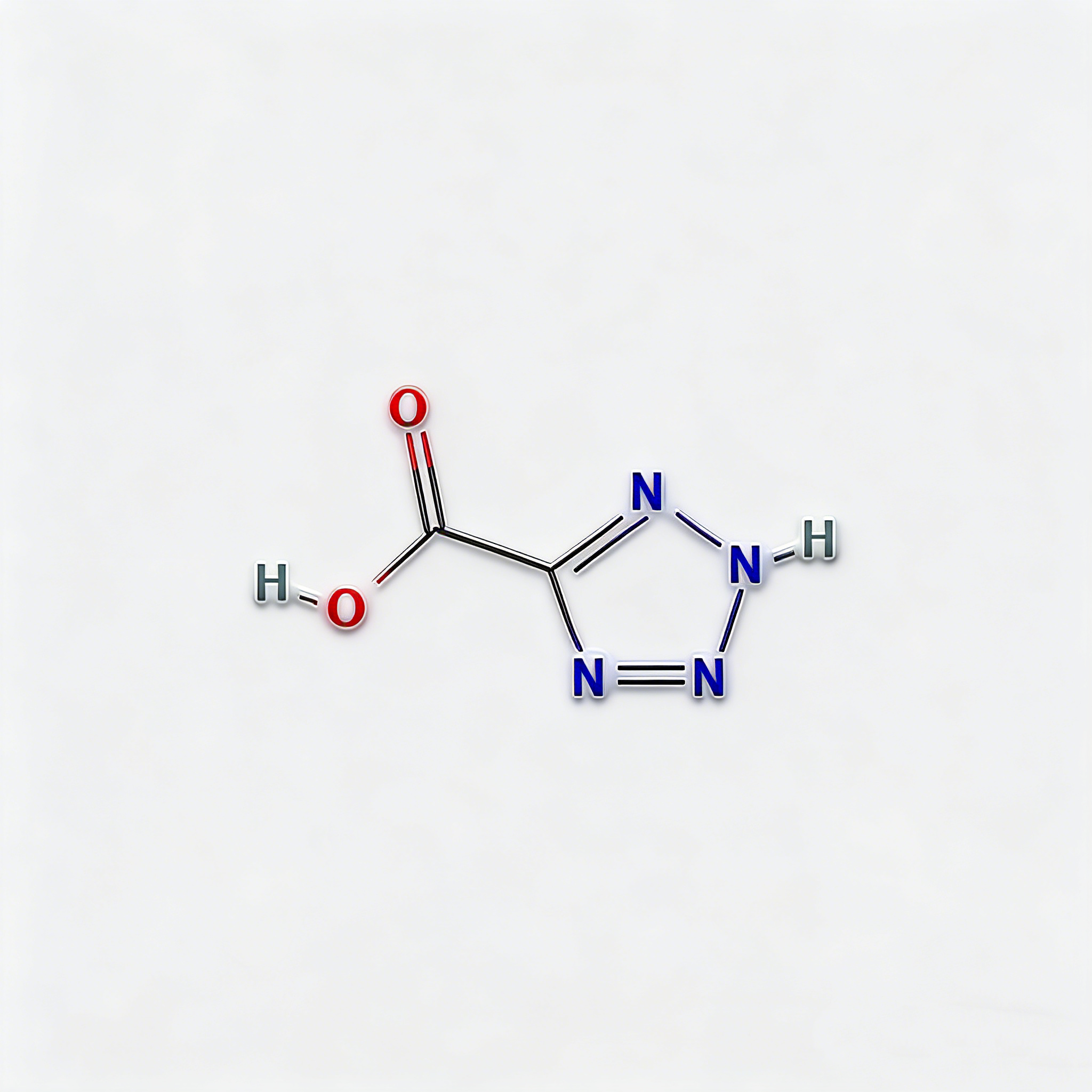
3-Bromo-1H-1,2,4-triazole (CAS 7343-33-1) is a strategically brominated five-membered heterocyclic scaffold engineered for regioselective functionalization in medicinal chemistry and agrochemical development. This molecule integrates a 1,2,4-triazole core—a privileged pharmacophore in kinase inhibitors, antifungal agents, and CNS modulators—with a bromine substituent at the C3 position. The C3-bromo handle enables efficient Pd-catalyzed cross-coupling (Suzuki, Buchwald-Hartwig, Sonogashira) or nucleophilic aromatic substitution, while the triazole ring provides dual hydrogen-bond donor/acceptor capability for enhanced target binding affinity. This intermediate is particularly valuable for constructing heterocyclic kinase inhibitors, antifungal azole analogs, and bioisosteric replacements for imidazole or pyrazole motifs where metabolic stability and solubility modulation are critical. Manufactured with purity ≥98% under controlled conditions, it supports drug discovery programs targeting fungal CYP51, B-Raf kinase, and adenosine receptor pathways.
Property
|
Product Name
|
3-Bromo-1H-1,2,4-triazole
|
|
CAS Number
|
7343-33-1
|
|
Chinese Name
|
3-溴-1H-1,2,4-三唑
|
|
Synonyms
|
1H-1,2,4-Triazole, 3-bromo-; 3-Bromo-1,2,4-triazole;
|
|
Chinese Synonyms
|
3-溴-1,2,4-三唑; 溴代三唑; 1H-1,2,4-三唑-3-溴; 3-溴代-1H-1,2,4-三唑
|
|
Molecular Formula
|
C₂H₂BrN₃
|
|
Molecular Weight
|
147.96 g/mol
|
|
Purity
|
≥ 98% (GC/HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Triazole Derivatives / Brominated Heterocycles
|
|
Appearance
|
White to Off-white Crystalline Powder
|
|
Melting Point
|
187.0 – 193.0 °C
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong bases to avoid triazole ring degradation.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Versatile building block for synthesizing pharmaceuticals, agrochemicals, and coordination complexes. Key intermediate in kinase inhibitor and antifungal agent development. Used in medicinal chemistry for bioisosteric replacement of imidazoles/pyrazoles and in materials science for functional materials synthesis.
|
|
Reactivity
|
The bromine atom at the 3-position is suitable for cross-coupling reactions (Suzuki, Buchwald-Hartwig, Sonogashira) and nucleophilic substitution. The N-H group (tautomeric) is available for alkylation/acylation. The triazole ring can act as a ligand for metal coordination
|