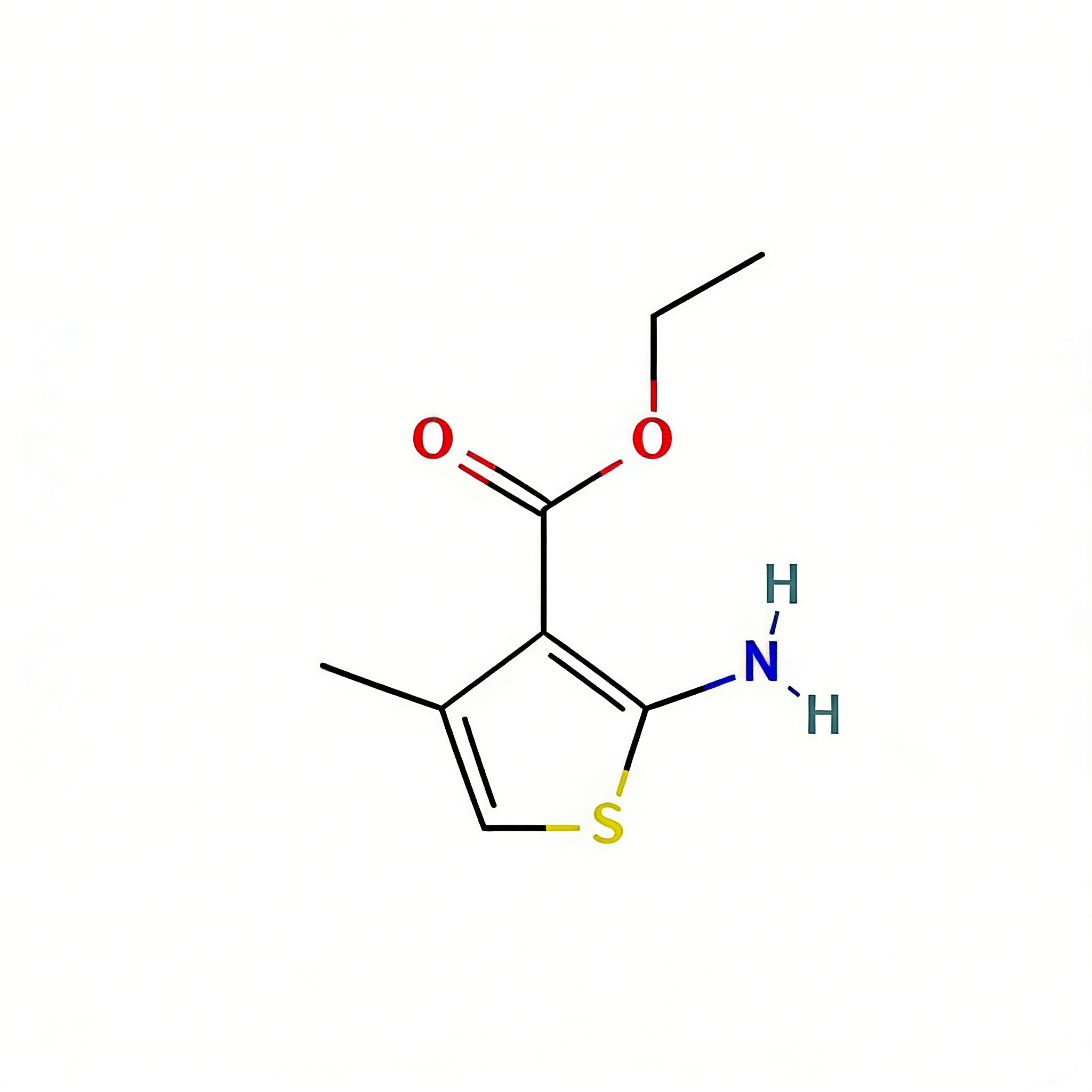
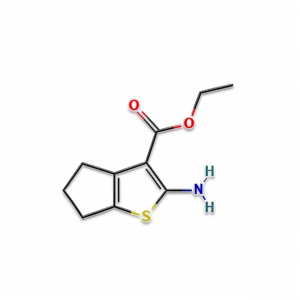
Ethyl 2-amino-4H,5H,6H-cyclopenta[b]thiophene-3-carboxylate
Ethyl 2-amino-4H,5H,6H-cyclopenta[b]thiophene-3-carboxylate (CAS 4815-29-6) is a strategically fused bicyclic thiophene derivative engineered for heterocyclic construction and medicinal chemistry applications. Distinct from monocyclic aminothiophene esters, this molecule features a cyclopentane ring fused to the thiophene core, creating a rigid 5,5-bicyclic architecture that enhances metabolic stability and target binding affinity. The classic 2-amino-3-ester substitution pattern facilitates efficient condensation cyclization with 1,3-dielectrophiles (e.g., formamidine acetate, urea) to construct fused thienopyrimidines, thienopyridines, and other polyheterocyclic systems prevalent in kinase inhibitors. The saturated carbocyclic ring provides conformational constraint and improved lipophilicity compared to aromatic benzo-fused analogs, while the ethyl ester moiety offers versatile handles for hydrolysis or transesterification. This intermediate is particularly valuable for constructing ATP-mimetic kinase inhibitors, CNS-penetrant scaffolds, and agrochemical actives where the tetrahydrocyclopentathiophene motif optimizes physicochemical properties. Manufactured with purity ≥98% under controlled conditions, it supports drug discovery programs requiring modular access to rigid sulfur-containing heterocyclic architectures.
Property
|
Product Name
|
Ethyl 2-amino-4H,5H,6H-cyclopenta[b]thiophene-3-carboxylate
|
|
CAS Number
|
4815-29-6
|
|
Chinese Name
|
2-氨基 -4H,5H,6H-环戊并 [b] 噻吩 -3-羧酸乙酯
|
|
Synonyms
|
Ethyl 2-amino-5,6-dihydro-4H-cyclopenta[b]thiophene-3-carboxylate
|
|
Chinese Synonyms
|
2-氨基 -4,5,6,7-四氢环戊并 [b] 噻吩 -3-羧酸乙酯; 氨基四氢环戊噻吩羧酸酯; fused 噻吩中间体; 乙酯类杂环化合物
|
|
Molecular Formula
|
C₁₀H₁₃NO₂S
|
|
Molecular Weight
|
211.28 g/mol
|
|
Purity
|
≥ 98% (GC/HPLC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Fused Thiophene Derivatives / Amino Ester Heterocycles
|
|
Appearance
|
White to Off-white to Pale Yellow Crystalline Powder
|
|
Melting Point
|
~113-117°C (estimated, literature reference recommended)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong oxidizing agents to avoid amino group oxidation or sulfur oxidation.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100g/bottle, 500g/bottle, 1kg/bottle, 5kg/drum, or customized according to customer requirements
|
|
Applications
|
Thienopyrimidine synthesis, kinase inhibitor scaffold development, CNS drug precursor research, agrochemical active ingredient precursors, heterocyclic library synthesis via condensation cyclization
|
|
Reactivity
|
Suitable for condensation with 1,3-dielectrophiles (cyclization), ester hydrolysis to acid, amino group acylation/alkylation, electrophilic aromatic substitution, and sulfur oxidation to sulfone/sulfoxide
|
|
Special Note
|
✅ Fused Bicyclic Scaffold—rigid 5,5-system enhances binding affinity vs. monocyclic thiophenes. 2-NH₂/3-COOEt motif enables rapid access to fused thienoheterocycles. Part of our fused thiophene building block series.
|

![Ethyl 2-amino-4H,5H,6H-cyclopenta[b]thiophene-3-carboxylate 4815-29-6](https://wealsunbio.com/wp-content/uploads/2026/01/4815-29-6.png)