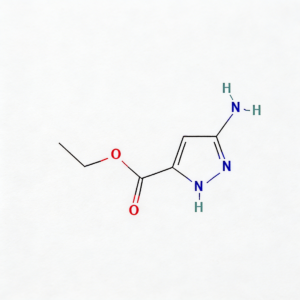
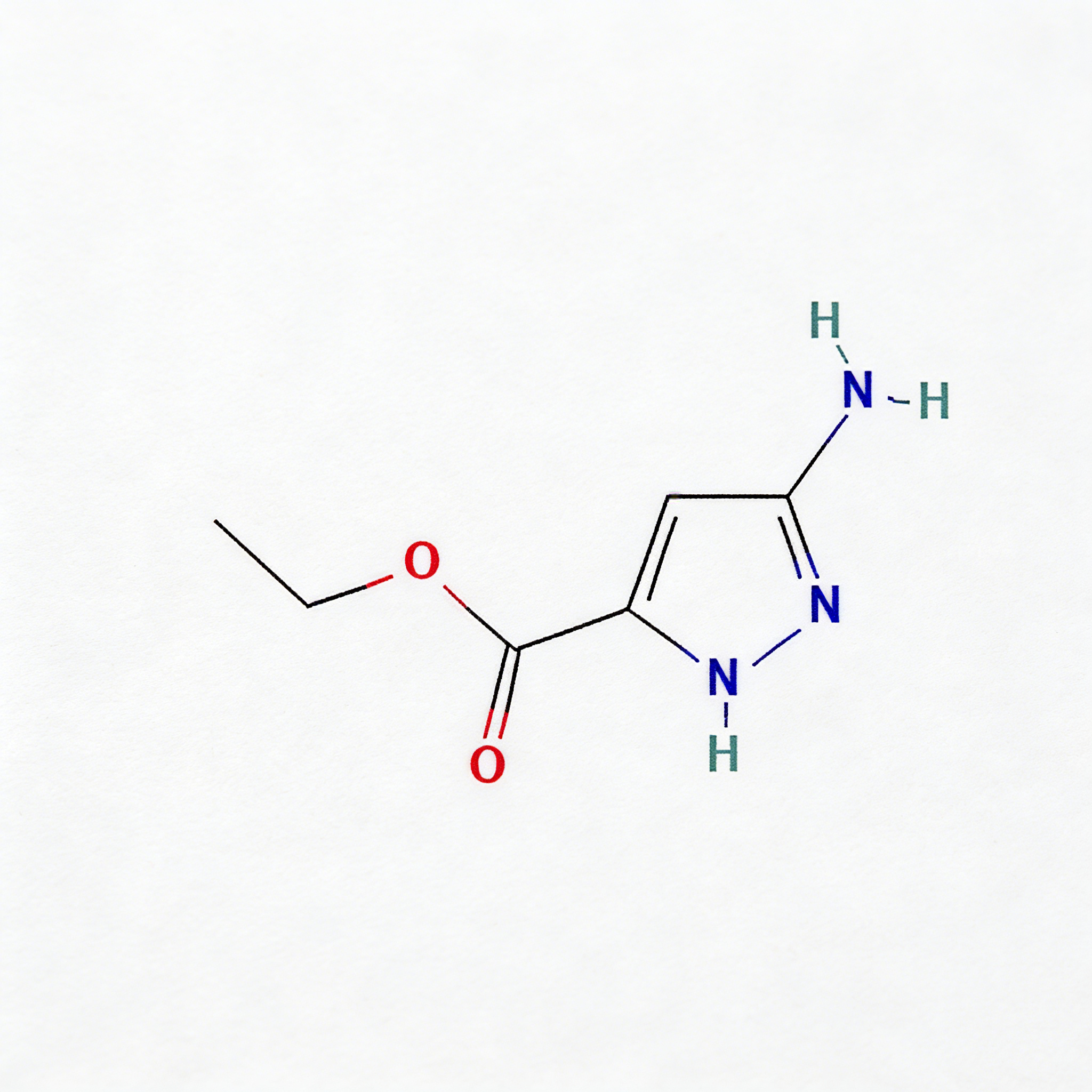
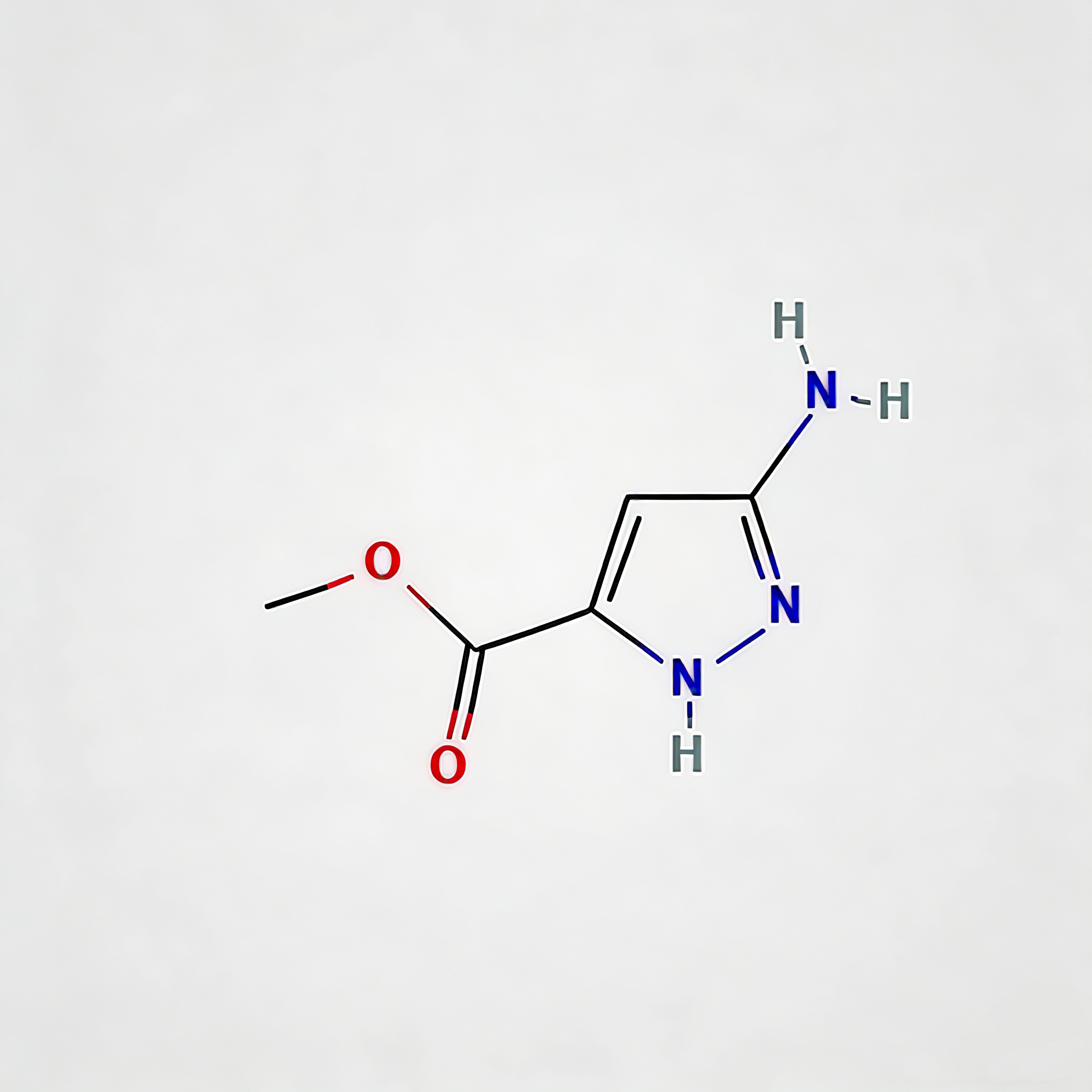
Ethyl 5-amino-1h-pyrazole-3-carboxylate
Ethyl 5-amino-1H-pyrazole-3-carboxylate (CAS 105434-90-0) is a strategically functionalized pyrazole building block for heterocyclic diversification in medicinal chemistry and agrochemical development. This molecule integrates three orthogonal reactive handles: a C5-amino group enabling electrophilic substitution or condensation cyclization, a C3-ethyl ester suitable for hydrolysis or amide coupling, and a 1H-pyrazole core that serves as a privileged scaffold in kinase inhibitors, anti-inflammatory agents, and CNS modulators. The electron-rich pyrazole ring—combined with the activating amino substituent—facilitates regioselective functionalization at C4, while the ester moiety provides a versatile anchor for peptide coupling or prodrug design. This intermediate is particularly valuable for constructing fused heterocyclic systems (e.g., pyrazolo[1,5-a]pyrimidines), kinase inhibitor cores targeting ATP-binding pockets, and agrochemical actives where the aminopyrazole motif enhances target selectivity. Manufactured under controlled conditions, it supports drug discovery programs requiring modular access to nitrogen-rich heterocyclic architectures.
Property
|
Product Name
|
Ethyl 5-amino-1H-pyrazole-3-carboxylate
|
|
CAS Number
|
105434-90-0
|
|
Chinese Name
|
5-氨基-1H-吡唑-3-羧酸乙酯
|
|
Synonyms
|
Ethyl 5-aminopyrazole-3-carboxylate; 1H-Pyrazole-3-carboxylic acid, 5-amino-, ethyl ester; 5-Amino-3-ethoxycarbonylpyrazole; C₆H₉N₃O₂ heterocycle; Ethyl 3-amino-1H-pyrazole-5-carboxylate
|
|
Chinese Synonyms
|
5-氨基吡唑-3-羧酸乙酯; 5-氨基-3-乙氧羰基吡唑; 氨基吡唑羧酸酯; 5-氨基-1H-吡唑-3-甲酸乙酯
|
|
Molecular Formula
|
C₆H₉N₃O₂
|
|
Molecular Weight
|
155.15 g/mol
|
|
Purity
|
≥ 98% (HPLC area normalization)
|
|
Product Category
|
Pyrazole Derivatives; Heterocyclic Building Blocks
|
| Appearance | Pale yellow to light brown solid |
|
Melting Point
|
117-121°C
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong acids to avoid ester hydrolysis or pyrazole ring protonation.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100mg/vial, 500mg/vial, 1g/bottle, 5g/bottle, or customized according to customer requirements
|
|
Applications
|
Kinase inhibitor synthesis, fused heterocycle construction (pyrazolo[1,5-a]pyrimidines), anti-inflammatory drug development, agrochemical active ingredient precursors, dye chemistry intermediates, prodrug design via ester modification
|
|
Reactivity
|
Suitable for electrophilic aromatic substitution at C4, condensation with 1,3-dicarbonyls to form fused rings, ester hydrolysis to acid, amide coupling, amino group acylation/alkylation, and pyrazole N1-derivatization
|
|
Special Note
|
Dual-reactivity scaffold—C5-NH₂ for cyclization/electrophilic substitution, C3-COOEt for coupling/hydrolysis. The 5-aminopyrazole motif is a key building block for ATP-mimetic kinase inhibitors. Part of our nitrogen-rich heterocyclic series.
|