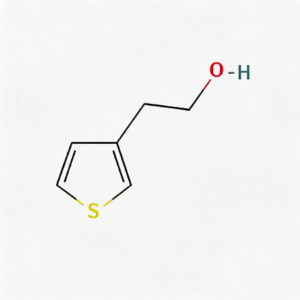
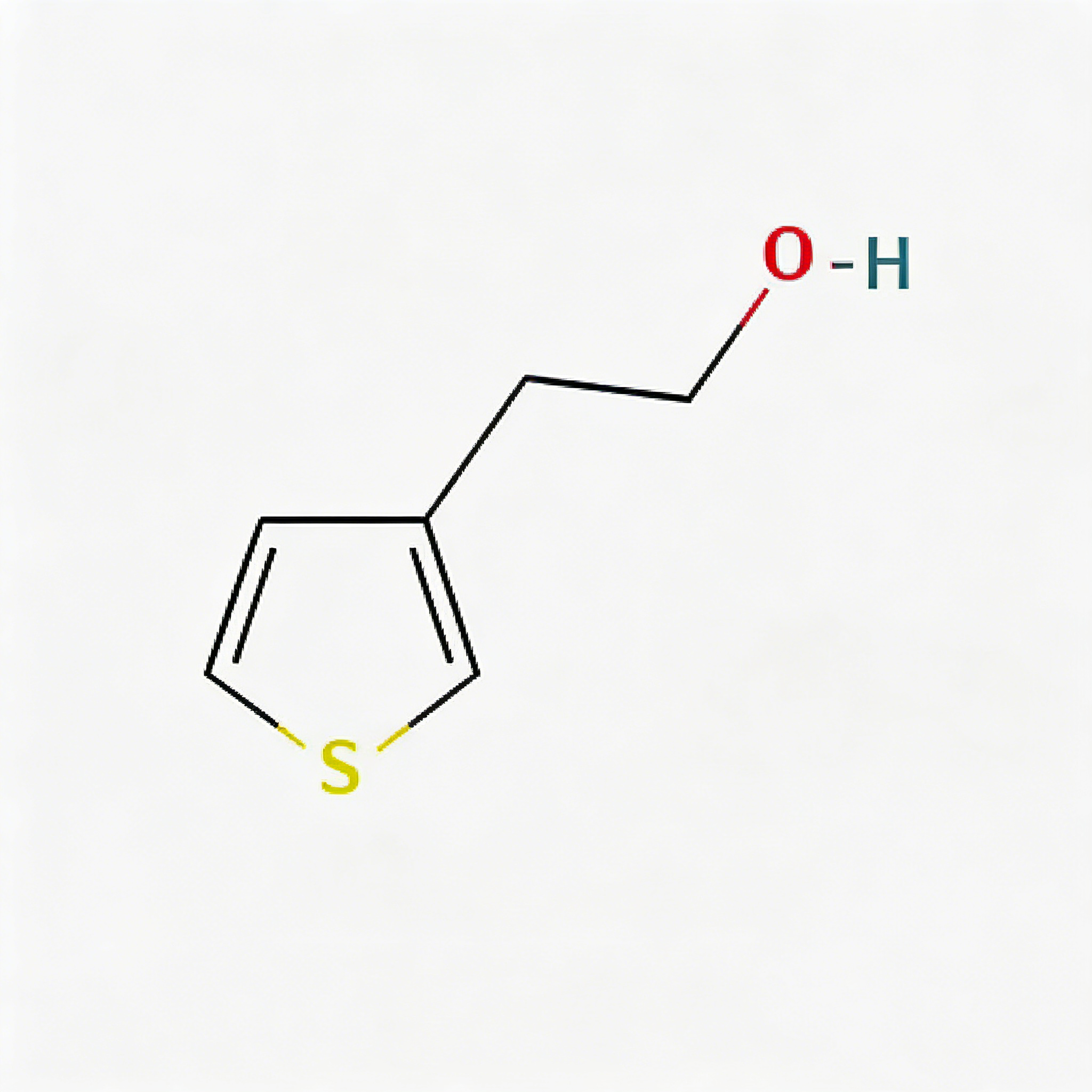
2-(Thiophen-3-yl)ethanol
2-(Thiophen-3-yl)ethanol (CAS 13781-67-4) is a strategically functionalized thiophene derivative engineered for pharmaceutical intermediate synthesis, ligand development, and materials science applications. This molecule integrates a 3-substituted thiophene core—a sulfur-containing five-membered heterocycle with excellent electron-donating properties and π-conjugation potential—with a 2-hydroxyethyl side chain at the C3 position. The primary alcohol terminus serves as a versatile handle for esterification, etherification, oxidation to aldehyde/acid, or conversion to halides for further cross-coupling strategies. The meta-like substitution pattern on the thiophene ring provides distinct electronic distribution compared to 2-substituted analogs, enabling unique binding orientations in target proteins. This intermediate is particularly valuable for constructing CNS-penetrant drug scaffolds, sulfur-containing ligands for metal coordination, and conductive polymer precursors where the thiophene-ethanol motif balances lipophilicity with synthetic flexibility. Manufactured with purity ≥98% under controlled conditions, it supports drug discovery programs requiring modular access to functionalized thiophene architectures with tunable physicochemical properties.
Property
|
Product Name
|
2-(Thiophen-3-yl)ethanol
|
|
CAS Number
|
13781-67-4
|
|
Chinese Name
|
2-(噻吩 -3-基) 乙醇
|
|
Synonyms
|
3-(2-Hydroxyethyl)thiophene; Thiophene-3-ethanol; 2-(3-Thienyl)ethanol; C₆H₈OS heterocycle
|
|
Chinese Synonyms
|
3-(2-羟乙基) 噻吩; 噻吩 -3-乙醇; 2-(3-噻吩基) 乙醇; 羟乙基噻吩
|
|
Molecular Formula
|
C₆H₈OS
|
|
Molecular Weight
|
128.19 g/mol
|
|
Purity
|
≥ 98% (GC area normalization)
|
|
Product Category
|
Pharmaceutical Intermediates / Thiophene Derivatives / Hydroxyalkyl Heterocycles
|
|
Appearance
|
Colorless to Pale Yellow Liquid
|
|
Boiling Point
|
~240-250°C at 760 mmHg (estimated)
|
|
Density
|
~1.15 g/cm³ at 20°C (estimated)
|
|
Refractive Index
|
n₂₀/D ~1.52 (estimated)
|
|
Storage Conditions
|
Store in a cool, dry, well-ventilated area away from light and moisture. Keep container tightly closed under inert atmosphere (nitrogen). Protect from strong oxidizing agents to avoid sulfur oxidation or alcohol degradation.
|
|
Shelf Life
|
24 months under recommended storage conditions
|
|
Packaging
|
100g/bottle, 500g/bottle, 1kg/bottle, 5kg/drum, or customized according to customer requirements
|
|
Applications
|
Pharmaceutical intermediate synthesis, ligand development for metal coordination, conductive polymer precursor research, agrochemical active ingredient precursors, alcohol derivatization (esterification/etherification/oxidation)
|
|
Reactivity
|
Suitable for esterification, etherification, oxidation to aldehyde/acid, conversion to halides (for cross-coupling), electrophilic aromatic substitution at C2/C5, and sulfur oxidation to sulfone/sulfoxide
|
|
Special Note
|
✅ Versatile Thiophene Building Block—primary alcohol enables diverse derivatization strategies. 3-substitution pattern offers distinct electronic profile vs. 2-substituted thiophenes. Part of our thiophene building block series.
|